Tomtor, a Treasury of "Rarities"
It was not too long ago that the industrial development of a country was judged from cast iron and steel production. The age of iron and coal is gone. Recent decades witness continuous growth of the use of so-called rare elements. It is related to the appearance and development of new technologies and new branches of industry. We buy complicated business machines, launch fireworks, or use cell phones without thinking of the fact that their production involves rare elements: samarium, neodymium, cerium, strontium, etc. At present, Russian industry is short of these elements in spite of their low demand, which is met by import, whereas ample reserves of rare elements have been explored and prepared for development. Their mining can ensure the independence on foreign rare elements for hundreds of years
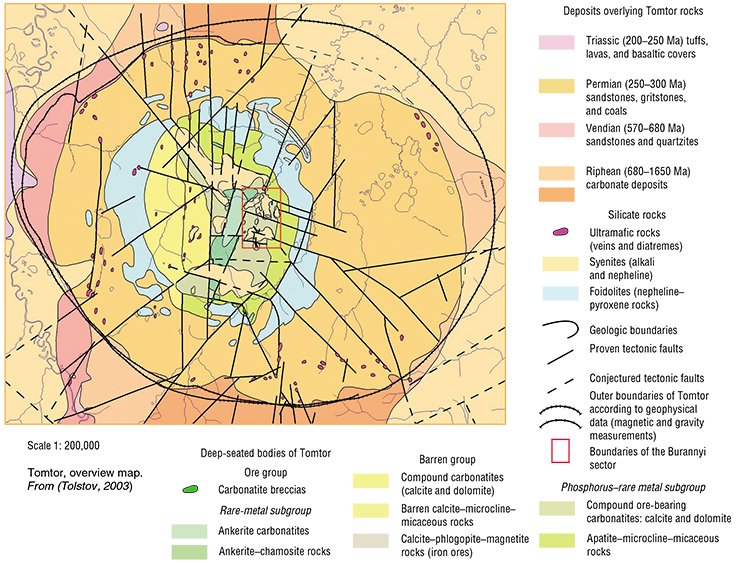
The Tomtor ore massif is located in northernmost Yakutia, 250 km from the Arctic Ocean coast. It was discovered in late 1950s by Leningrad geologists S.A. Gulin and E.N. Erlikh. It is worth noting that Sergei Gulin figured Sergei Gurin, the protagonist in the widely popular O. Kuvaev’s novel Territory, describing the life of Soviet geologists in that epoch.

The main feature of the massif is a giant field of compound ores. At present, the State Register of Reserves accounts ten elements, but the hallmarks of Tomtor are niobium, yttrium, scandium, and lanthanides, present in great abundance and highly concentrated.
The main minerals of Tomtor ores are niobium pyrochlore and rare-earth phosphates. Usually, their concentrations are no greater than 1–5%, whereas in Tomtor they are major components. They constitute 10 to 80% of the rocks! Actually, the rocks are niobium–REE concentrate. The production of such concentrate from common ores demands giant concentrating mills, and in Tomtor, it is given by Nature itself.
Unfortunately, although the unique ore deposit has attracted attention since its discovery, its remoteness from industrial regions by thousands of kilometers hampered its development. Even now, fifty years after the discovery, the present and future of the giant rare element deposit are obscure.
Does Russia need Tomtor in the light of dramatic changes in the raw-material base and the structure of global economy?
The rare-element epoch
The term rare elements is loose. It has been conventionally used since the times when most of them were rare not even with respect to their industrial use rather than abundance in the Earth’s bowels. This “fashionable” group was visited by over 50 elements, including those currently utilized as major structural materials in things of daily use: titanium, molybdenum, tin, etc.
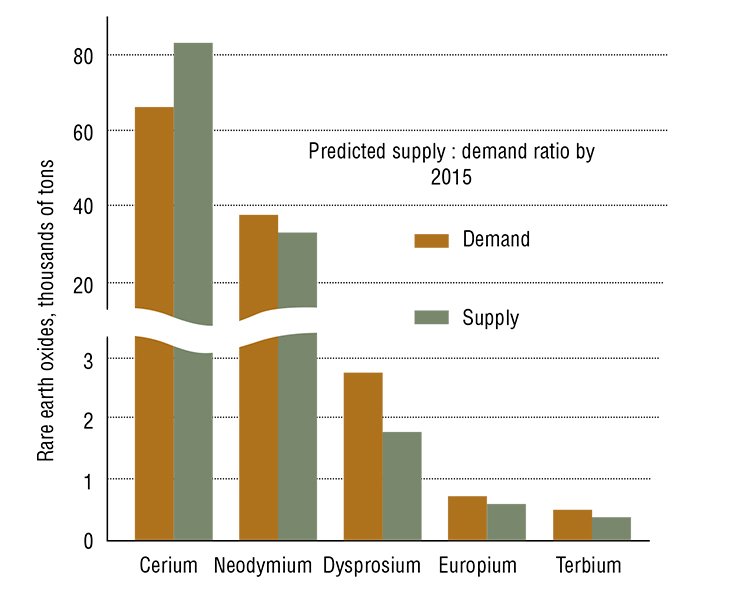
Russia ranks second to China in proven rare earth reserves and certainly first in resources. It has the largest deposits of niobium and rare earths (Tomtor and Chuktukon) and tantalum (Katugin). Most of the reserves of these elements are enlisted in the State Register as associated components of complex ores (mainly apatite). As a result, over 80,000 tons of rare earth oxides are deducted from the balance annually, whereas no more than 15000—25000 tons are extracted in the form of commercial products. Thus, the amount of rare earth elements falling to waste is twenty times as large as the demand of Russian industry. However, Russia has no production facilities for converting such waste, and the construction of new ones is economically unsound in the modern context
Presently, rare elements include 35 members: 17 rare-earth elements (scandium, yttrium, lanthanum, and lanthanides) and rare metals (tantalum, niobium, lithium, beryllium, zirconium, etc.)

Experts indicate that nowadays the consumption of rare metals is a more important index of industrial development of a country than cast iron or steel. It is not an overstatement to say that all aerospace, nuclear, ultra-high-temperature, ultramagnetic, ultralight, ultrahard, and ultrastrong engineering materials are manufactured on the base of rare elements or with their use. At present, many rare metals that were out of use for long bring into existence fields of modern industry: solar energy, magnetically levitated ultrahigh-speed vehicles, infrared optics, optoelectronics, computing technologies, and much more.

Magma differentiation, giving rise to carbonatites, requires specific conditions. The chamber must not be too deep; otherwise, pressure hampers carbonatite formation. Neither must it be too close to the surface, to prevent their fast cooling. Then carbonatite and ultramafic magma intrude to subsurface strata and form veins. Generally such carbonatite bodies are no more than 1 km in diameter, but the diameter of the unique central body of Tomtor is about 6 km.
Carbonatite massifs occur in the most permeable areas, which are sites of branching, junction, or cross of differently directed faults. Carbonatites have the largest ore contents of all assemblages of ultramafic alkali rocks. The products of subsurface alteration (weathering) of carbonatites are even richer. Just they contain commercial types of ores with a large set of raw materials: niobium, phosphorus, iron, and rare-earth elements
A paradoxical feature of the global rare element market is the relatively low cost of cerium-group elements (lanthanum and cerium), whose ample resources exist mainly in China, and the very high cost of other elements, including yttrium-group rare earths and scandium, which restricts their utilization.
The consumption of rare elements per capita varies from country to country manifold and even by orders of magnitude. The consumption of rare elements per one ton of steel or per capita is an index of the economic maturity of a country. For the majority of less developed countries, including Russia, this index is several times (or by an order of magnitude for some rare metals) less than in advanced countries.
Giant of giants
The scientific and technical revolution started in the USSR later than in advanced countries. At the beginning of 1980, USSR lagged far behind in the consumption of rare metals. For example, the per capita consumption of niobium was one-eighth as large as in Italy and one-seventh as in West Germany. The consumption of zirconium was one-ninth as in Japan, and so on. The lion’s share of these elements was consumed by the Soviet arms industry.
Prospecting work was done in Tomtor in the middle 1980 by Yakutskgeologiya, and I found pyrochlore–monazite–crandallite ores, previously unknown. The discovery of enormous reserves of these ores and their unique ore content changed the view of the deposit dramatically, and it was acknowledged to be among the greatest in the world. However, the mineral treasure of Tomtor became needless in the perestroika epoch
Later, the situation worsened, especially in the last quarter of the 20th century. The Soviet rare-metal industry disintegrated, and its enterprises fell into different countries: Kazakhstan, Russia, and Estonia. Apparently, the broken economic links cannot be restored as they were.
Presently, limited amounts of rare-earth ores are mined only at the Lovozero enterprise (Murmansk oblast). The ore is converted to loparite concentrate, which is processed at the Solikamsk magnesium plant, Perm Krai. Both enterprises are privately owned, and their capital goods are worn out to a significant extent. Thus, a decrease in production is expected.
The demand for rare metals in Russia is almost entirely met by import. The shortage of rare earths in Russia has been increasing since 2012. Presently, it is about 300 tons/year. This fact may retard the development of Russian high-tech industry in the decade to come.
The production of rare elements can be increased at the Lovozero enterprise, but it would concern only light lanthanides, whose liquidity is poor. A fundamental change of the situation demands operation of new unique enterprises able to meet market needs.

Russia has enormous mineral reserves of rare elements, gravitating mainly to northern and eastern regions: the Kola, Maimecha–Kotun, East Sayan, Sete-Daban, and Udzha carbonatite provinces are the base of the ore potential of iron, phosphorus, and rare elements.
Even in comparison to these deposits, Tomtor is a true leader. It is known that natural concentrations of rare elements are broadly variable, usually within few parts per million (scandium), per 105 or per 104 (tantalum), or per thousand (niobium, cerium, and lanthanum). Commercially significant concentrations are several times higher. The contents of rare element oxides in Tomtor ores astonish: 1—24% niobium, 0.1—3.5% yttrium, 0.01—0.15% scandium, and 1—39% other rare-earth metals.
Compare these figures with the famous Aracha field in Brazil, where about 90% of the global niobium production is mined. The contents of niobium oxide in its ores range within 1.0—2.5% with next to no secondary rare earths. Ores of another large rare-earth mining district, Bayan-Obo in China, contain up to 10% rare metals, but no niobium. The assessment of Tomtor reserves done in 1999 in accordance with the standards of the State Reserves Commission shows that the boundary value of niobium oxide content, at which the ore is considered commercial, is 3.5%, above the mean concentration in Aracha. The reserves of such commercial ores in the Burannyi sector alone are 42,700,000 tons.
Thus, Tomtor is now the first rare-metal giant in the world in the resources and concentrations of niobium and rare earths. Its development would support the required increase in the production of the whole range of rare-earth products and highly liquid secondary products: aluminum and titanium oxides.
The nascence of Tomtor
How did the unique rare-earth ores of Tomtor arise? There are three major hypotheses of their origin: deposition in lakes (sedimentary origin), transformation of effused lava flows (effusive–sedimentary origin), and concentration of insoluble heavy rare elements in stepwise subtraction of rock-forming elements in the course of successive oxidation and reduction.
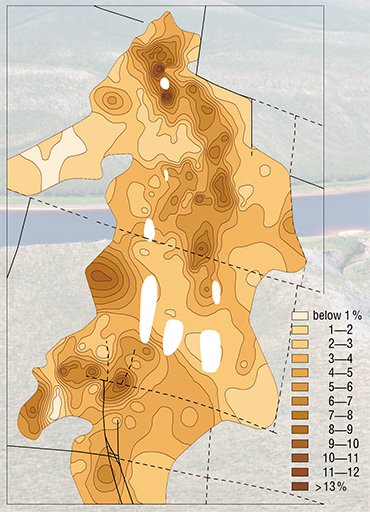 According to the last hypothesis, put forward by Lapin and Tolstov (1991; 1993), the main ore formation stages included weathering of initially ore-bearing carbonatites followed by redeposition and structural–compositional alteration of parent rocks under nonequilibrium conditions of the water/rock system (epigenetic transformation). At the last stage of genesis, the ores were buried under sediments, which protected them from erosion.
According to the last hypothesis, put forward by Lapin and Tolstov (1991; 1993), the main ore formation stages included weathering of initially ore-bearing carbonatites followed by redeposition and structural–compositional alteration of parent rocks under nonequilibrium conditions of the water/rock system (epigenetic transformation). At the last stage of genesis, the ores were buried under sediments, which protected them from erosion.
At last, the following scenario emerges. Tomtor itself was forming as a deep magma chamber for long, about 200 Ma (within 600 to 350 Ma BP). Probably, it included two or three independent steps. The resulting intricately built massif appeared on the day surface no earlier than the Devonian, when primitive fish appeared in seas.
At that time, the climate in what is now Arctic Yakutia was similar to tropical Africa. Under those temperatures, rocks experienced deep transformation, particularly influencing ore-bearing compound carbonatites of heterogeneous composition. As a result, a 300-m thick iron–phosphate cap formed above the massif. Iron oxides, hematite and limonite, hued it brownish-rusty.
Thewn soluble carbonate rocks were washed out during karstification, and the initial mass of carbonatites decreased by 50 to 90%. In this way, ore components rich in niobium and rare earth accumulated.
Large depressions formed in central areas of the massif experiencing karstification at the Carboniferous/Permian boundary. In addition, tectonic movements were caused by repeating earthquakes occurring in the Carboniferous. All these processes redistributed the loose matter. Rocks were eroded from elevated areas and deposited in depressions. That was how three local sectors arose: Northern, Burannyi, and Southern. They were extraordinarily enriched in ore minerals with clear niobium–rare-earth predominance.
Later, in the Early Permian, the depressions were filled by freshwater reservoirs, where organic sediments accumulated. Thus, oxidative settings gave way to reduction. Iron (III) and manganese (IV) were converted to soluble compounds with oxidation rate II. Their subtraction further increased the concentrations of insoluble species of niobium, phosphorus, and rare earths.
The Permian lacustrine depressions were then filled by gravels and sands with coal interbeds. They formed a geochemical barrier, which prevented the ores from washout in the Triassic. In the Jurassic, the vast Northern Sea covered the entire Tomtor area and gave rise to marine deposits exceeding 100 m in thickness.
This ore formation pattern appears to be typical for all carbonatite massifs of central type (Lapin and Tolstov, 1991). Hence, indications for search for unique ores in redeposited weathering crusts can be developed for known massifs of the type in Russia or abroad.
Everything is usable
The unique Tomtor ores are a natural mixture of concentrates. Therefore, their mechanical processing is complicated. It is based on hydrometallical ore benefication, which allows production of good-quality concentrates with a high degree of valuable component extraction.
President of Russia V.V. Putin, Annual Address to the Federal Assembly, Kremlin, Moscow, December 12, 2012
A two-step process was developed at the Fedorovsky All-Russia Research Institute of Mineral Raw Materials, Moscow, and the Federal State Research and Design Institute of Rare Metal Industry with considerable contributions of experts from the Institute of Chemistry and Chemical Technology, Krasnoyarsk. It involved alkaline decomposition with phosphate withdrawal followed by treatment with nitric or hydrochloric acid with chlorination of the solid residue and extraction of rare-earth elements. This method allows utilization of up to 75% of ore components.
The uniqueness of the method is that it yields nearly 1.2 tons of commercial products from one ton of ore. How can it be? The ore processing offers, in addition to rare metal materials, by-products: alumina, titanium, and even trisodium phosphate, the base of detergents. Other useful by-products include alkaline aluminate solution, hydrochloric acid, and compounds of barium and strontium, which can be recovered at certain steps of the process and recycled.
The mode of occurrence of redeposited Tomtor ores allows their mining in a quarry, whose initial capacity is estimated to be 10,000 tons of dry ore with subsequent increase to 100,000 tons/year. Note that niobium–rare earth ores are radioactive owing to the presence of uranium, thorium, and potassium-40. However, the radioactivity at the surface of the deposit is relatively low, 2 to 22 µR/h, which is within the limits of natural radiation background.
The development of the deposit is expected to damage areas no farther than 2.5 km from the quarry. Most elements in the ore are in the form of hardly (if at all) water-soluble compounds, which can migrate in the oxidized and/or colloidal/suspended states only in the flood season.
Therefore, the main hazardous factor of environment pollution with toxic radioactive elements and heavy metals is the wind-blown dispersal of mineral particles from the quarry and waste dumps. Submeridional winds are predominant in the deposit area all round the year. This can be taken into account in the outlining of the shape and size of the quarry to minimize the dispersal of dangerous and radioactive dust.
Finally, the necessary surveying and prospecting work in the Tomtor rare-earth deposit, discovered about 50 years before, was completed, and an appropriate technology was elaborated for ore concentration. Now the deposit is ready for mining. If it is put into operation, the capital assets will pay back within five years. It should be mentioned that the high capital : output ratio of Russian mining projects hampers their execution without state support in the form of special licensing, subsurface management, and taxation. Tomtor is no exception in this regard
The remoteness from densely populated regions is no insurmountable problem at present. Diamond fields are mined 80 km west of the deposit. Diamond enterprises Almazy Anabara and Nizhnelenskoe operate there and process millions of cubic meters of rock per sluicing season, whereas the expected ore mining volume in Tomtor is about ten times less. In addition, ore can be delivered to the mouth of the Anabar River and then to Krasnoyarsk by water.
Already the first step of development will be able to cease the import of niobium and rare metals. Then, Russia will become a prominent rare metal market participant, ranking with China and Brazil.
It is of no less importance that the execution of this project will be in accordance with the political aspirations of Russia in the exploration of northern Siberia and whole Arctica. It will reanimate these vast poorly explored regions of Russia. With the start of Tomtor development, the cluster model of industrial development can be used in Yakutia, which will involve the Popigai lonsdaleite (hexagonal diamond) deposit, located 150 m west of Tomtor, and hydrocarbon deposits.
Anyway, Tomtor development has no alternative with the increasing economic development of Russia and its need for rare elements. This project must be part of the program of growth and innovational development of up-to-date branches of the Russian industry and a prerequisite for its success.
References
Kravchenko S. M., Belyakov A.Yu. Novichok sredi gigantov. Priroda. 1992. No.4. S. 50—55.
Kravchenko S. M., Belyakov A.Yu., Kubyshev A.I., Tolstov A.V. Skandievo-redkozemel’no-ittrievo-niobievye rudy – novyj tip redkometall’nogo syr’ja // Geologija rudnyh mestorozhdenij. 1990. T. 32. No. 1. S. 105–109.
Lapin A.V., Tolstov A. V. Novye unikal’nye mestorozhdenija redkih metallov v korah vyvetrivanijakarbonatitov // Razvedka i ohrana nedr. 1993. No. 3. S. 7—11.
Tolstov A. V. Problemy geologo-jekonomicheskoj ocenki Tomtorskogo mestorozhdenija: Materialy Mezhdunar. simpoz. «Strategija ispol’zovanija i razvitija mineral’no-syr’evoj bazy redkih metallov v ХХI veke». M.: 1998. S. 135—137.
Tolstov A. V. Problemy ocenki i osvoenija niobij-redkozemel’nyh rud Tomtorskogo mestorozhdenija: Materialy XIV Mezhdunar. soveshh. po geologii rossypej i kor vyvetrivanija. Novosibirsk, 2011. S. 656—660.
Erlikh E. N. Novaja provincija shhelochnyh porod na s-v Sibirskoj platformy // Zap. VMO. 1964. T. 93. No. 6. S. 682—693.







