From Olfaction in Fish to Regenerative Medicine
When describing the ability of nerve cells to adapt, neurobiologists use the term neuroplasticity, which is ingrained in the biological literature. This term does not mean, however, that neurons are malleable like plasticine; they can transform, but within a certain structural and functional framework, beyond which the cell dies. By studying neuroplasticity, researchers, on the one hand, solve puzzles of nature and, on the other hand, try to use this phenomenon to address issues of human health
Olfaction is the most ancient sense organ. We perceive smells with the help of chemoreceptor cells, which are sensitive to chemical substances. These cells are bipolar neurons, and, like many neurons, they have one axon, a long projection going to the brain, and one dendrite, a short projection. The dendrite of the chemoreceptor cell extends into the external environment with its sensors, or cilia (flagella), whose membranes contain membrane-bound receptor proteins.
In many animals, chemoreceptor cells have developed throughout the evolution a highest degree of specialization; in fact, they reached the physical limit of the discrimination capacity of the olfactory system and are able to unerringly identify individual molecules and send information to the brain. Especially important are the chemical signals that determine the eating, defensive, and reproductive behavior. Moreover, a recent discovery shows that olfaction is used to evaluate more subtle biological characteristics such as social status, physiological condition, and other personalized data about individuals of one’s own or other species.
How do these nerve cells acquire such a high specialization in the course of individual development? What are the resources underlying the reliability of their work? These questions are important not only for the study of the complex behavior of animals but also for some issues of human health, such as combating Alzheimer’s disease and transplantation issues.
Yellowfin Baikal sculpin: sexual behavior based on smell
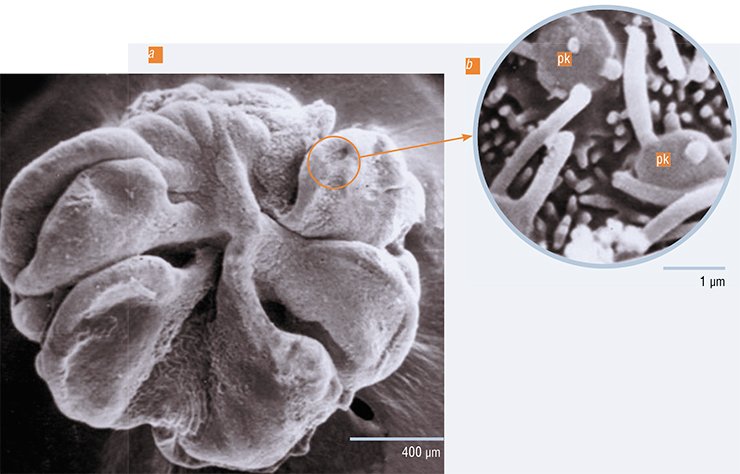
The neuroplasticity phenomenon involves both functional cell changes and structural ones, which can be seen under an electron microscope. To identify clear morphological, i. e. ultrastructural, criteria for high or low sensitivity of receptor cells, scientists need a good model. Such a model was found in the chemocommunicative behavior of the yellowfin Baikal sculpin (Cottocomephorus grewingkii).
 It turned out that when the spawning season comes, the males of these fish acquire a very high sensitivity to the female sex pheromone. In the female’s ovary, mature eggs are stored in the ovarian fluid, which keeps them, for a relatively long time, ready for fertilization. The ovarian fluid is rich in biologically active compounds called pheromones, i. e. substances that cause certain neuroendocrine and behavioral responses in an animal of the same species as the individual that secretes them.
It turned out that when the spawning season comes, the males of these fish acquire a very high sensitivity to the female sex pheromone. In the female’s ovary, mature eggs are stored in the ovarian fluid, which keeps them, for a relatively long time, ready for fertilization. The ovarian fluid is rich in biologically active compounds called pheromones, i. e. substances that cause certain neuroendocrine and behavioral responses in an animal of the same species as the individual that secretes them.
The pheromones not only attract the male yellowfin sculpin to the female but also synchronize (with the female!) the maturation of his sexual products. More detailed studies have shown that one of the key components of the female pheromone—17-dihydroxyprogesterone steroid—is effective at a very low concentration of 10-11–10-13 M.
The corresponding experiment looks very impressive: if you take the roe from one female, put it into one hundred milliliters of water and then add one drop of the solution to an aquarium with a male ready for spawning, then, even without seeing the object of his adoration, he will immediately begin to demonstrate sexual behavior. Driven by the smell, the male rolls over and his body begins to vibrate, excreting sexual products “in vain.” Females also respond to the pheromones which males release into water with urine.
That is, a very small portion of biologically significant molecules can radically change the behavior of the fish. Notably, in the spawning period, sculpins have pronounced sexual dimorphism. The male’s head becomes carbon-black, and his fins become lemon-yellow: this color helps attract a female and withstand the tough competition for her. In this respect, females are less attractive: like most fish species, they almost do not change their color during spawning; i. e. the male does not attach particular importance to the color of the sexual partner. However, the chemical signals produced by the ready-to-spawn female are of paramount importance.
Thus, during the spawning season, the olfactory cells of male yellowfin sculpins somehow switch from the perception of food signals, which are typical of the inter-spawn period, to pheromonal ones, without which these fish cannot propagate themselves.
From olfactory to secretory
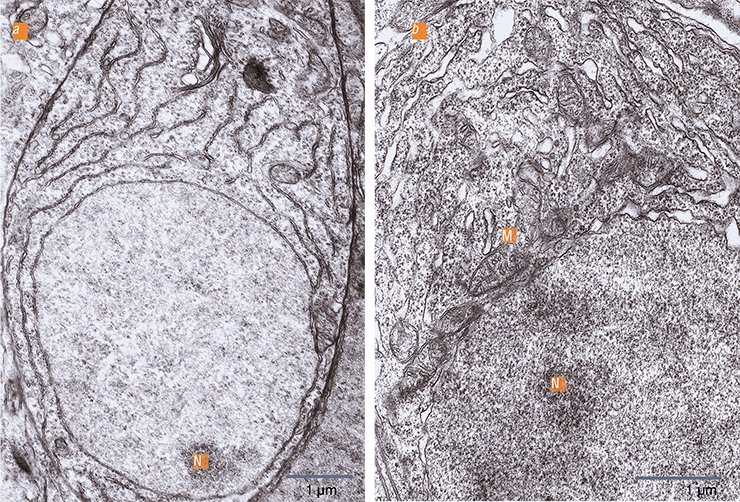
When the chemoreceptor cells of yellowfin sculpins become sensitive to pheromones, they begin to show ultrastructural signs of increased metabolism. There is an increase in nuclear cytoplasmic interactions: the nucleolus becomes active; the nuclear membrane shows an increased density of pores; near which the mitochondrial “power plants” concentrate. Consequently, in the cytoplasm there is an increase in ribosomes, i. e. “molecular machines” for protein synthesis. The chemoreceptor cell dendrites are observed to have clearly structured microtubules, which are elements of the cytoskeleton, i. e. the transport system of the neuron. This is important since adaptive changes in the cells must affect the need in the corresponding enzyme systems which are urgently delivered through the microtubules from the cell body to the receptive area. Thus, we found morphological parameters that accompany the adaptation of the olfactory cells to the perception of very low concentrations of chemical agents.
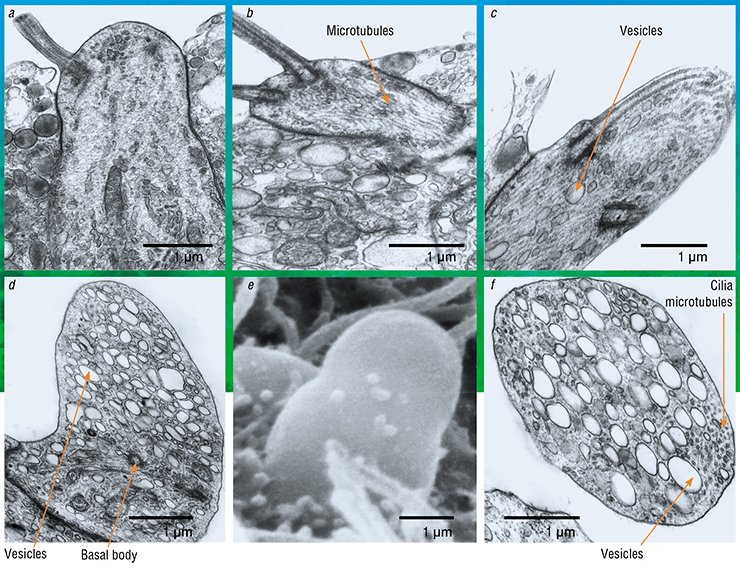
We must admit that these structural changes are not fundamentally different from those in the other neurons of the central nervous system, when they are at work. These changes are commonly referred to as reactive properties of the neuron, which, given an optimal functional load, exhibits structural signs of stress, i. e. adaptation to the stimulus.
SIMILAR TO IMMUNE CELLSThe evidence of immune principles in the functioning of olfactory cells comes from some of the important properties of the latter. Like lymphocytes, olfactory cells are monospecific and have a great diversity (polymorphism) of the genes that encode odorant-binding receptors (Buck and Axel, 1991). On top of this, there are two important facts which have been obtained quite recently. Analysis of the human genome revealed that promoters (the place where the RNA synthesis begins), which controll the expression of olfactory receptor genes and of the major histocompatibility complex proteins, also have a very high point polymorphism (Ignatieva et al., 2014).
In addition, there is another unexpected circumstance in support of the still little-understood evolutionary parallelism of the olfactory and immune systems: last year, German scientists discovered olfactory and taste receptors in the membranes of immune cells (Malki et al., 2015)
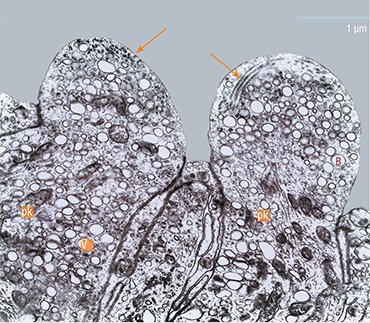
However, the other changes are not typical of sensitive cells. When male sculpins switch to parental behavior, i. e. protecting the roe, some receptor cells reveal, against the background of enhanced protein synthesis, the activation of the Golgi apparatus, i. e. a system of intracellular membrane structures in which various substances mature and accumulate to be later transported to their destination. The cells clearly show a directed transport of secretory vesicles to the top of the cell where they are embedded into the membrane, releasing their contents into the olfactory mucus.
During the transformation, the chemoreceptor cells lose their surface receptor apparatus, i. e. sensitive cilia. Their cytoskeleton is immersed into the top of the dendrite, and the cell appears to lose the chemoreception ability. That is, the cells cease to be olfactory... now they are secretory!
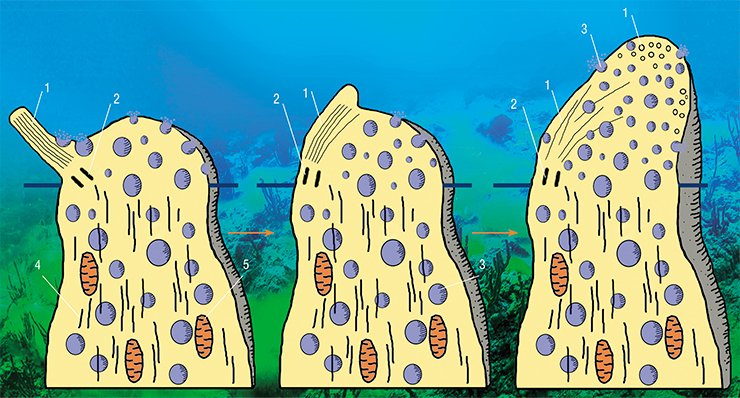
This does not fit into the traditional understanding of the functional purpose of olfactory neurons. From the earliest works (Schultze et al., 1856) to the present day, it has been generally believed that the top of the chemoreceptor cell is always specialized only in chemoreception. Furthermore, it is well known that neurosecretion in any nerve cell, with some exceptions, goes through the axon, which transmits information to another cell, not through the dendrite.
It is true that some authors described elements of morphological change that appear to suggest the reorganization of the olfactory cells into the dendritic neurosecretion mode. The key elements of these reorganizations—bubbles with a light content in different quantities—were seen in the dendrites and receptor cell tops in many animals and even in humans. However, these studies revealed no substantial increase in the secretory function of the Golgi apparatus, which would be accompanied by substantial restructuring of the cell top. Therefore, unfortunately, the bubbles found in the olfactory cells have so far attracted little attention.
Beyond spawn season
Dendritic neurosecretion occurs in fish not only in the spawning period. One can see similar ultrastructural reorganizations of olfactory cells if a fish is subjected to a prolonged odor impact with nontoxic water-soluble substances; we used a mixture of amino acids and peptides.
We assumed that the stimulated chemoreceptor neurons begin to produce not a membrane-bound, but a water-soluble form of proteins (Klimenkov et al., 2015), which bind the odorant molecules directly in the olfactory mucus, providing an adaptive loss of smell: if a fish has long been exposed to an odor, it stops feeling the smell.
Although this is a fascinating hypothesis, it remains unclear whether the cells can switch from the synthesis of a membrane-bound form of receptor proteins to that of a secreted form while maintaining their specificity, because it is still unknown how to collect the water-soluble form of the receptors. It is possible that, during dendritic neurosecretion, neurons may switch to the synthesis of some other proteins or short neuropeptides. Such an interpretation gives rise to the “oligogene” hypothesis of the encoding of receptor proteins, according to which olfactory cells can express, at different stages in their development, not one but several genes that encode the odorant receptors (Mombaerts, 2004).

It should be noted that olfactory cells with dendritic neurosecretion are morphologically similar to hypothalamic neurosecretory cells, whose “job” is to synthesize and release substances of different chemical nature, which take part in information processing and ensure neuroplasticity. Currently, researchers admit that, along with amino acids and monoamines, these substances include neuropeptides secreted by both the axons and dendrites of nerve cells (Leng and Ludwig, 2008). Neuropeptides may act not only in the synapse. Their half-life is rather long; they diffuse into the extracellular space and can affect more distant targets (Son et al., 2013). Therefore, in addition to the quick-acting neurotransmitters, neuropeptides are considered as a major means of chemical communication in the brain. Thus, the dendritic neurosecretion phenomenon, which is observed in some types of central nervous system cells, is perhaps not an extraordinary property of olfactory neurons either.
Compensatory neurogenesis and medicine
It was found in experiments that, during a prolonged exposure to an odor, some olfactory cells undergo neurodegenerative changes and die. This is preceded by damage to the mitochondria in these cells and increased formation of free radicals, which is observed during the development of many pathological processes. We have to make a paradoxical conclusion: even completely harmless odorous substances, when taken in excess amounts, affect the cell as a stress factor.
We hypothesized that in such conditions, there should be compensatory neurogenesis processes in the sensitive epithelium. Indeed, the intensity of proliferative activity in the experimental fish was higher by a factor of 2.6 than in the control group. That is, the formation of new cells in animals’ sensor apparatus can be activated not only after the transection of the olfactory nerve and mechanical or acute toxic impacts, as we believed, but also during an enhanced odor stimulation and the subsequent degenerative changes.
This phenomenon is very important from a practical point of view. Now there is a great concern about the sources of undifferentiated neural cells which could be cultivated and used as a biological “glue” to treat traumatic lesions of the nervous system. The most important requirement for such a “glue” is its genetic compatibility with the cells of the individual who needs the treatment. Addressing this problem encounters many challenges, including ethical issues. Baikal sculpins could provide exciting material not only for understanding their biology and mechanisms of adaptation to the chemical factors of the environment but also for using this knowledge to address some issues of regenerative medicine. The olfactory epithelium is the only available source of autologous undifferentiated neural cells which can be used for transplantation in the treatment of the nervous system diseases (Viktorov, 2006).
There is a theory to support this idea: the structural and functional properties of the olfactory epithelium are not different in fish and in humans, and it is necessary to conduct studies on mammals. The epithelium should be treated with an odorant in a water-soluble, not volatile form, whereby the solution is injected into the nasal cavity. In this case, neurons should be better activated, which would lead to their more intense degradation, with subsequent compensatory activation of the stem cell division and formation of a pool of undifferentiated neural cells which have potential applications in medicine. From a practical point of view, taking such a biopsy is quite simple.
Another aspect is that the olfactory system is involved in the development of a number of socially significant neurodegenerative diseases such as Alzheimer’s, Parkinson’s, etc. It is known that dementia (impairment of the higher functions of the brain), which is usually due to aging, is accompanied by a decrease in the quantity of chemosensitive cells and an olfactory dysfunction. If we find out how to noninvasively stimulate neurogenesis at the level of the peripheral olfactory analyzer, the lost cells could at least be partly replenished. We should not exclude the possibility of the positive effect of the increased influx of odors on the central structures of the brain with which olfactory cells are associated. We can even suggest that this phenomenon lies at the core of the therapeutic effects of aromatherapy, which was born many centuries ago in the East.

In conclusion I would like to quote our colleague, a prominent expert in neurobiology, Professor N. S. Kositsyn (Institute of Higher Nervous Activity and Neurophysiology, Moscow), who said: “In these studies, Lake Baikal is more than a unique endemic-rich lake, it is a methodological approach that gives us natural models to investigate complex issues of medicine as well as biology.”
References
Buck, L., Axel, R. A novel multigene family may encode odorant receptors: a molecular basis for odor recognition // Cell. 1991. V. 65, N 1. P. 175—187.
Ignatieva, E. V., Levitsky, V. G., Yudin, N. S. et al. Genetic basis of olfactory cognition: extremely high level of DNA sequence polymorphism in promoter regions of the human olfactory receptor genes revealed usingthe 1000 Genomes Project dataset // Front Psychol. 2014. V. 5. Article 247. Published online.
Klimenkov, I. V., Kurylev, A. V, Kositsyn, N. S. et al. Olfactory Receptor Cells’ Dendritic Neurosecretion Phenomenon // World neurosurgery. 2015. V. 83, N 3. P. 278–279.
Leng, G., Ludwig, M. Neurotransmitters and peptides: whispered secrets and public announcements // J. Physiol. 2008. V. 586, N 23. P. 5625–5632.
Malki, A., Fiedler, J., Fricke, K. et al. Class I odorant receptors, TAS1R and TAS2R taste receptors, are markers for subpopulations of circulating leukocytes // J. Leukoc. Biol. 2015. V. 97, N 3. P. 533–545.
Mombaerts, P. Odorant receptor gene choice in olfactory sensory neurons: the one receptor-one neuron hypothesis revisited // Curr. Opin. Neurobiol. 2004. V. 14, N 1. P 31–36.
Schultze, M. Uber die Endigungswise des Geruchsnerven und der Epithelialgebilde des Nasenschleimhaut // Monatsber Deut Akad. Wiss. Berlin. 1956. Bd. 21. S. 504—514.
Son, S. J, Filosa, J. A, Potapenko, E. S. et al. Dendritic peptide release mediates interpopulation crosstalk between neurosecretory and preautonomic networks // Neuron. 2013. V. 78. N 6. P. 1036–1049.
Viktorov, I. V., Savchenko, E. A., Ukhova, O. V, et al. Multipotent stem and progenitor cells of the olfactory epithelium // Bull. Exp. Biol. Med. 2006. Oct. 142(4). P. 495-502.
Based on the materials of joint research with Cand. Sci. (Bio.) N. P. Sudakov (Irkutsk Scientific Center of Surgery and Traumatology; Irkutsk Scientific Center, Siberian Branch, Russian Academy of Sciences; Irkutsk State University), A. V. Kurylev (Irkutsk State University), Cand. Sci. (Bio.) M. V. Pastukhov (Vinogradov Institute of Geochemistry, Siberian Branch, Russian Academy of Sciences, Irkutsk), and Dr. Sci. (Bio.), Prof. N. P. Kositsyn (Institute of Higher Nervous Activity and Neurophysiology, Russian Academy of Sciences, Moscow)