Lake Baikal: Earthquakes Can Be Predicted
On December 9, 2020, an earthquake occurred near Lake Baikal, a strong yet not dangerous one. It happened in the famous Proval Bay, where in 1862, astonished people saw a huge plot of land collapse and sink under water. To a depth of several meters, as it turned out. The last earthquake came in handy as it gave Irkutsk scientists the opportunity to get evidence proving they are on the right track in a very ambitious project to build a model that allows one to predict future earthquakes
The path to building a new earthquake prediction model has been a long one. When my colleagues told me what they were up to, I did not believe them. Thousands of scientists worldwide had been struggling with the problem of accurate prediction of earthquakes, and Japanese researchers remained firmly in the lead. Therefore – so I thought – the chances of my colleagues were vanishingly small. As it turned out, they were right and I was wrong. I am happy that I did put a stop to that work although I could.

So, from the point of view of fundamental science, it is a major breakthrough, the one our respected leaders have dreamt of. But since they are not scientists and have every right not to understand this, we ought to help them. I would also like to bring these ideas to a broad audience, especially high school students.
Point of origin: The Nuclear Project
The road was long. At the end of World War II, the Americans detonated nuclear bombs in Hiroshima and Nagasaki. I was born in 1939, and until 1944, I lived with my mother and grandmother in Novosibirsk, in evacuation. That year, my mother was ordered to go to the United States, where my father was on temporary duty.
When I heard on the radio that the Americans had detonated nuclear bombs, I asked my father: “Does Stalin have such a bomb?” My father said, “Of course, he does, son.” In fact, Stalin did not have a bomb back then, not until 1949. The Soviet Union had very little uranium, not enough even for one bomb. Uranium was found later in the occupied Germany and Czechoslovakia. The works were classified to the utmost degree; even saying the word “uranium” was forbidden.
Lavrenty Beria was in charge of the Soviet nuclear project. This man knew how to give orders. All Soviet geological expeditions were to carry a strongbox with a revolver and a radioactivity counter, and all of these expeditions received a secret order reminding everyone to look for uranium. Several powerful agencies were set up for a single purpose of finding uranium. One could not have done all this enormous work had it not been for the contribution of the most talented, most prominent Soviet scientists. When uranium was found, some of these scientists would carry out some unclassified studies in their free time.

We should begin our story by citing the names of the world-renowned Soviet radiogeochemists Viktor Cherdyntsev and Pavel Chalov.
I apologize to the readers for boring them with many unexciting names. Please have patience. Homer’s Iliad gives the names of all the heroes who came to war with Troy. These names say little to us, but they were very important to the ancient Greeks.
Below I write about uranium isotopes and about the phenomenon discovered by Cherdyntsev and Chalov. It is very difficult to explain all this to a general reader, even to a scientist who works outside that field. It took me myself a whole year to understand it. Well, may God help us!
Uranium “clock”
Very long time ago, the Earth came into being and turned into a solid body. It was a nuclear explosion, so almost all the elements were radioactive; over billions of years, almost all of them became nonradioactive. Perhaps, it is only uranium and thorium, the last and heaviest ones in the periodic table, that could have survived to this day. After billions of years, uranium isotopes with atomic masses of 238, 235 (this one is needed for a bomb), and 234 still remain radioactive.
 In 1931–1944, Viktor CHERDYNTSEV worked as a laboratory assistant at the Leningrad Radium Institute at the department headed by Academician Vladimir Vernadsky. In 1935, after another arrest of his father, Cherdyntsev was exiled from Leningrad to Tashkent “as a socially dangerous element.” After a petition by Vernadsky and his deputy Khlopin, the exile decision was revoked. Upon his return to Leningrad, he continued to work at the Radium Institute.
In 1931–1944, Viktor CHERDYNTSEV worked as a laboratory assistant at the Leningrad Radium Institute at the department headed by Academician Vladimir Vernadsky. In 1935, after another arrest of his father, Cherdyntsev was exiled from Leningrad to Tashkent “as a socially dangerous element.” After a petition by Vernadsky and his deputy Khlopin, the exile decision was revoked. Upon his return to Leningrad, he continued to work at the Radium Institute.
During the war, he lived in evacuation in Tatarstan and worked in the Chistopol region as the head of the expedition of the Radium Institute and the head of a radiological laboratory. He conducted radiological studies at oil fields. Having summarized the outcomes of these studies, he prepared and defended a dissertation for the degree of Candidate of Physical and Mathematical Sciences in the autumn of 1943.
In 1944–1950, he worked as deputy director of the Institute of Astronomy and Physics, Academy of Sciences of the Kazakh Soviet Socialist Republic (Alma-Ata), where he organized the first nuclear laboratory in Kazakhstan. In February 1946, he defended a dissertation for the degree of Doctor of Sciences on the topic “The Theory of the Origin of Atomic Nuclei.” In 1946–1960, he combined his work at the institute with chairmanship at the Department of Experimental Physics, Kazakh State University, where he organized a specialty in nuclear physics and set up a problem-oriented laboratory for the study of nuclear processes in nature and in cosmic rays.
Together with Pavel Chalov, his student and colleague at the Kazakh State University, he discovered the effect of separation of uranium isotopes, having received a priority certificate of discovery No. 163 dated March 27, 1954.
Pavel CHALOV, war participant, in the Red Army since December 1941 and in field forces since April 1942. Awarded battle honors. Graduated from the Kazakh State University (1951) and successfully completed a postgraduate program at the same university (1954)
Uranium‑238 continues decaying to this day. This element is abundant in terms of mass, but its decay rate is very low. It takes 4.5 billion years for uranium‑238 to decay by half. It is believed that the Earth appeared in its (more or less) modern form about 4.5 billion years ago. Radioactivity becomes a very important tool in the hands of researchers – imagine that every decayed atom can be measured with a very simple instrument, i. e., an ionization chamber. By the way, the first, yet not very accurate, determination of the Earth’s age was made by Arthur Holmes back in 1927.
All the other uranium isotopes appeared later; therefore, we focus only on two of them, 238 and 234. The half-life of uranium‑234 is only 250,000 years. It must have disappeared from rocks long ago, but it does not do so because it constantly emerges from uranium‑238. I is interesting and obvious that the radioactivity of uranium‑238 and –234 can only be the same. (Radioactivity is the number of decays per unit of time per unit mass. Radiochemists measure everything in units of radioactivity rather than in grams; they probably wish to make things more difficult for us poor laymen. They say that in uranium, the uranium–uranium ratio (uranium‑234/238) is usually 1. This is some Martian language, some hard-to-understand professional lingo, but we will keep trying.)
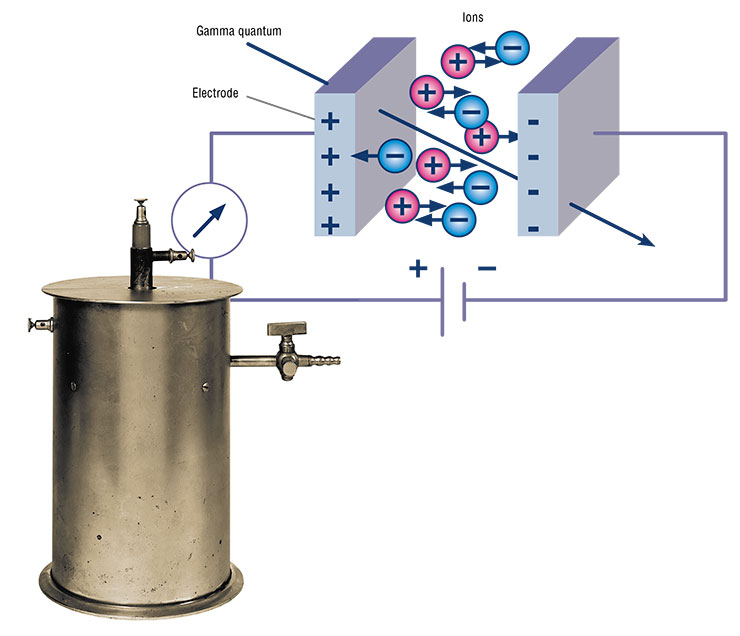
Every high schooler knows that the different isotopes of chemical elements do not differ at all from one another in terms of chemical properties, because their electron shell is the same, but the nuclei can be different.
Cherdyntsev and Chalov were, of course, searching for uranium deposits to be used in nuclear bombs, but they were genuine scientists, so they did not rest in their free time but measured the uranium–uranium ratios in a great number of rock and groundwater samples. As expected, the uranium–uranium ratio in rocks was 1. However, to the scientists’ complete amazement, some samples of suspended matter from well water showed a ratio much greater than 1. The record for uranium‑234/238 was 18! No one in the world had seen that before.
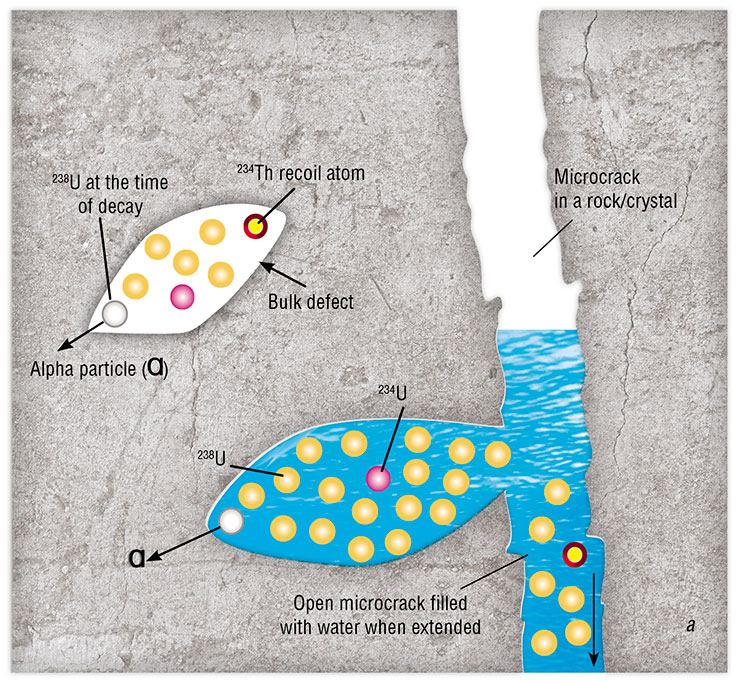
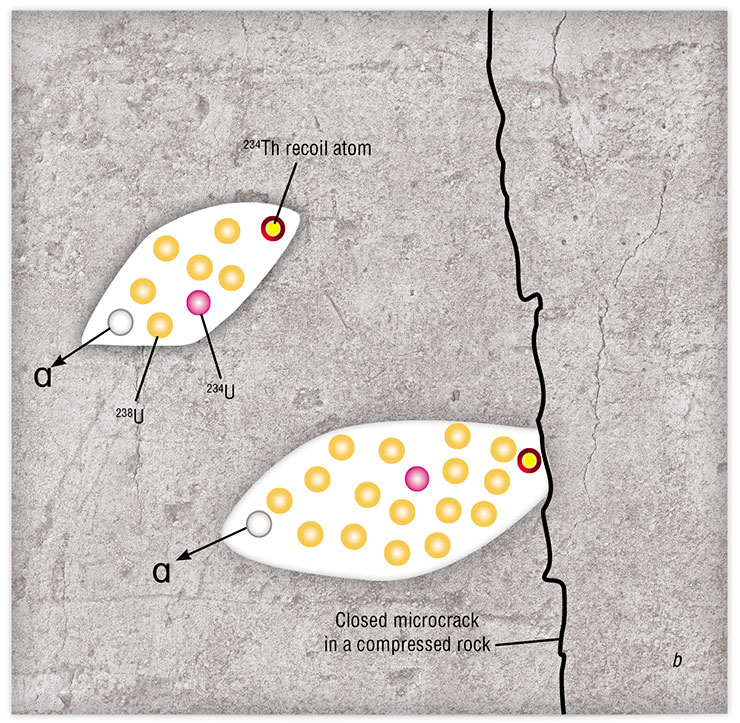
Cherdyntsev explained this phenomenon as follows: when a uranium atom decays, it leaves behind a track in the rock, i. e., a nanosized cylinder filled with water. He realized that this cylinder should contain the still highly radioactive newborn atom of uranium‑234, which emerges as a result of the decay of uranium‑238. Groundwater flowing around the track washes out the uranium, and this water gets into the sampler and then into the radioactivity detector and the ionization chamber.
Cherdyntsev and Chalov described their experiments in a language very easy to understand; it is no coincidence that their description, written in Russian, soon became very popular worldwide for approximate determinations of the age of groundwater and rocks in the range from 10,000 to 500,000 years. This art has long been lost. For example, to identify the radioactivity pulses of uranium‑234 and –238, they simply used tissue paper of different thicknesses.
Point of application: Lake Baikal
Now I have to digress from Chalov and Cherdyntsev and return to the city of Irkutsk near Lake Baikal. To the time when I was director of the Limnological Institute. At the beginning of the wild 1990s, one was free to do anything, and a huge number of good foreign scientists, especially American ones, came to Baikal. No wonder – for the first time, Baikal became fully open to foreigners whereas under the Soviet rule, it was virtually closed due to the secrecy regime.

The next key figure in my story is the recently deceased Prof. David Edgington, one of these scientists, who came from the Great Lakes WATER Institute (Wisconsin Aquatic Technology and Environmental Research Institute), Milwaukee, an elderly man back then. At that time, we were vigorously exploring Lake Baikal, especially its bottom sediments, and I understood very little in those things. Edgington and another American scientist, Steve Colman, explained to me what the uranium equilibrium was; they published the first paper on the uranium–thorium dating of Baikal sediments and dated the sediments associated with time intervals very important for understanding the evolution of our sacred lake, from 10,000 to 150,000 years ago. It turned out that using the same uranium‑234/238 ratio, one can not only date the age of the sediments but also get an idea of atmospheric precipitation at the center of Asia.
A few years passed by. A new man came to work at my institute, Evgeny Chebykin, back then still a student; Chebykin proved to be a man of an extremely rare nature – an excellent analytical chemist. At that crazy time, we managed to acquire a very expensive analytical instrument, i. e., a plasma-excited quadrupole mass spectrometer, an ICP-MS in the Martian language. I somehow came across a prospectus of that instrument; having examined it, I realized that the instrument could be used to measure uranium ratios not by radioactivity, a very difficult and very slow procedure, but in a matter of minutes (instead of several days) using the technique applied by Cherdyntsev and, oddly enough, by Edgington and Colman.
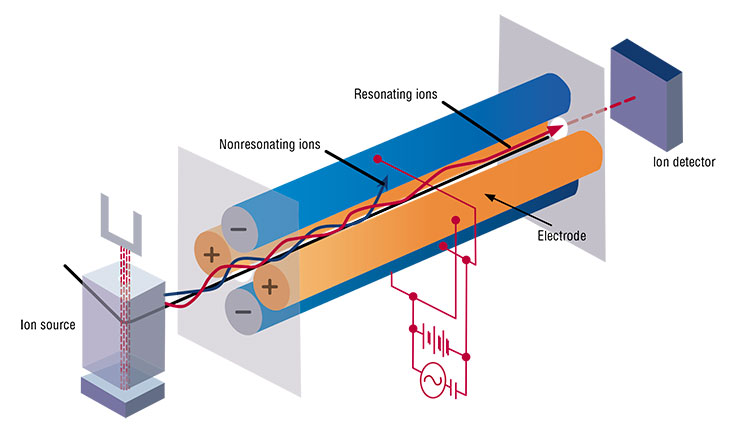
It was unusual – I did not have to beg the instrument out of Moscow officials in the hope of using it later in a hit and miss fashion; I ordered it purposefully to measure the uranium–uranium ratio (which must have bored you already). It was fabulous – we first arrived at the idea, and only after that we got the instrument in our laboratory. I am going to mention the ICP-MS frequently in my story because this instrument can simultaneously measure the concentrations of almost all the elements in the periodic table and is so sensitive that it can measure a vanishingly small concentration of uranium‑234.
Chebykin quickly mastered our ICP-MS and, to my surprise, all the sophisticated techniques of uranium–uranium dating. The whole world was already using the Cherdyntsev–Chalov method; the techniques had been described in the literature, but the ICP-MS analysis is an exceptionally delicate procedure, which can only be accomplished by analysts with a capital A, all of whom are exceptionally ill-tempered. С’est la vie.
 Edgington and Colman were able to measure the uranium–uranium ratio at only four or five points throughout the entire length of the 10‑meter core while Chebykin dated the core at points in 2‑cm increments with a resolution of 200 years over an interval of 10,000–140,000 years. It would be completely impossible to perform a core analysis with such a high resolution by the ancient radiometric method. But this is not the tale yet; the major story lies ahead.
Edgington and Colman were able to measure the uranium–uranium ratio at only four or five points throughout the entire length of the 10‑meter core while Chebykin dated the core at points in 2‑cm increments with a resolution of 200 years over an interval of 10,000–140,000 years. It would be completely impossible to perform a core analysis with such a high resolution by the ancient radiometric method. But this is not the tale yet; the major story lies ahead.
Soon after, a very good physicist suddenly appeared at our institute. He was kicked away – this is in the nature of things by us – from the Novosibirsk Institute of Solid State Chemistry and Mineral Resources Processing, Siberian Branch, USSR Academy of Sciences, for inventing an original technique for aerodynamic separation of nanometer-sized industrial diamonds. Very soon he joined our project and made an invaluable contribution to it. Unfortunately, he also passed away not long ago and left Chebykin an orphan; the poor man now has no one to talk to.
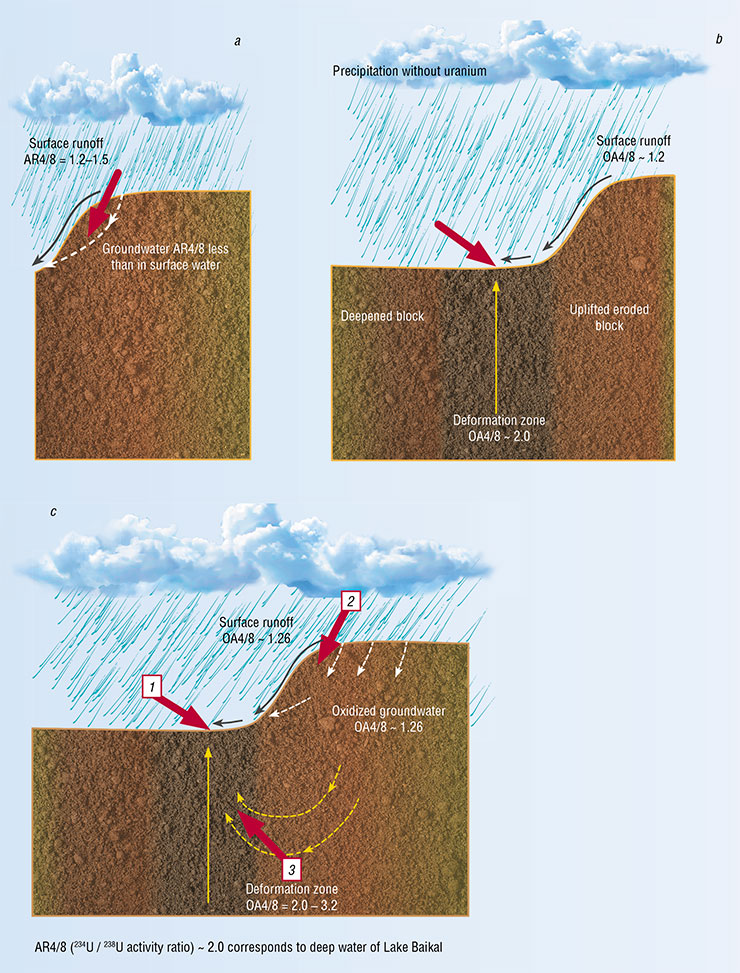
Evgeny Goldberg quickly refined the Cherdyntsev–Chalov model. The refinement seemed small, but it was in fact the most important brick in the observations of uranium–uranium hydroisotope responses to earthquakes. According to Goldberg, the past-flowing groundwater does not wash nonequilibrium uranium out of the tracks – for the sediment to be carried away, it must be crushed first. What could be the source of this abrasion in deep underground horizons, where there are neither people nor agate mortars?
 Goldberg did not have the time to ultimately prove his theory, but Chebykin began to work in a partnership with a very competent geologist Sergei Rasskazov, who is well-known for his studies of earthquakes at the Institute of the Earth’s Crust, Siberian Branch, Russian Academy of Sciences, also located in Irkutsk Akademgorodok. Rasskazov proved to be of great help already by familiarizing himself with the sensitive radiochemical and radiogeological studies mentioned above, although he never took any interest in these disciplines before.
Goldberg did not have the time to ultimately prove his theory, but Chebykin began to work in a partnership with a very competent geologist Sergei Rasskazov, who is well-known for his studies of earthquakes at the Institute of the Earth’s Crust, Siberian Branch, Russian Academy of Sciences, also located in Irkutsk Akademgorodok. Rasskazov proved to be of great help already by familiarizing himself with the sensitive radiochemical and radiogeological studies mentioned above, although he never took any interest in these disciplines before.
The recent Irkutsk earthquake made it possible to test Goldberg’s model professionally. Rasskazov and Chebykin have been carrying out persistently, for many years, hydroisotope monitoring in the vicinity of Lake Baikal; they somehow determined its tectonic state and put forward a hypothesis about the locations of the epicenters of future earthquakes. The currently available experience is still insufficient to accurately date and localize the predicted earthquakes, but it is clear that the guys are on the right track. They will tell you more about this.
So, now I give the floor to Sergei Rasskazov. I assure you this is a breakthrough achievement far above the world level. It has opened the path to accurate earthquake prediction, and in a decade, this path will be covered.
More often than not, seismologists only shrug their shoulders, “A strong earthquake happened again…” and move on to describing its consequences. Earthquake predictions are rare, but the world practice does know a few examples. There are more than 600 harbingers of strong earthquakes, but they are, in most cases, not regular. The harbingers may warn of one impending catastrophe but be useless in warning about another. The cause-and-effect relationships between the various effects emerging during the preparation of earthquakes remain behind the scenes.
Cherdyntsev and Chalov theoretically substantiated the effect of an increase in uranium‑234/238 activity ratios (AR4/8) in circulating underground waters as a result of seismogenic crustal deformations. In the 1970s, Chalov organized AR4/8 monitoring (as well as monitoring of other isotopic indicators) in underground waters of the Northern Tien Shan, but ironically, not a single earthquake occurred in that area for 1.5 years. Bad luck.

We are now doing what Chalov was unable to do. In the 1970s, they had to sorb enough uranium in order to measure AR4/8 by alpha-counting. In the case of low uranium content in fresh water, they used up to 300 liters, sorbing uranium with activated carbon. Today, we can make accurate measurements of a wide range of elements using the ICP-MS in only 2 ml of water, and we measure AR4/8 by isolating uranium from a half-liter plastic bottle of water.
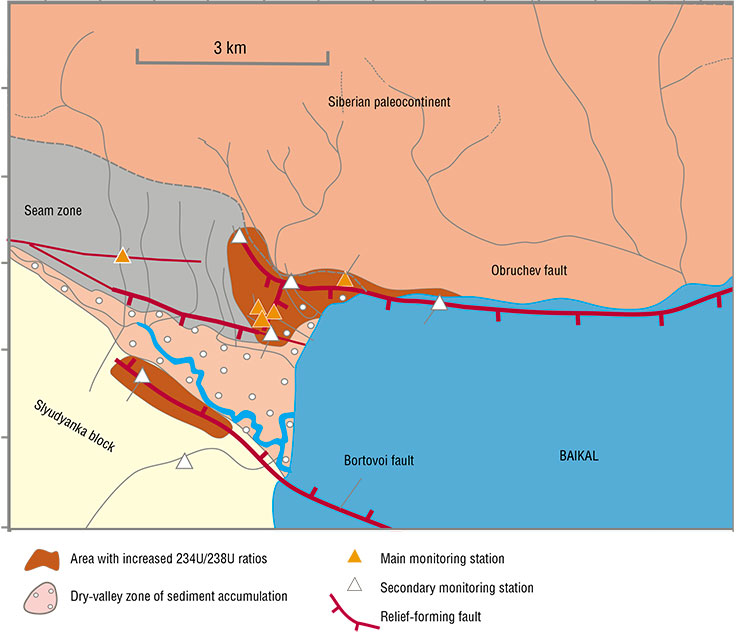
In order to organize a seismic prediction test site, we chose the most (geologically) sensitive area to earthquakes at the western tip of South Baikal, in the village of Kultuk. Before that, we collected water samples from 43 springs and wells in that village in order to identify and select six stations for ongoing observations in different seismically active faults. Over eight years of monitoring, we gained experience in recording the responses of uranium concentrations and AR4/8 to the preparation of weak and strong earthquakes in South Baikal, and by 2020, we had come to tracking possible seismic scenarios.
In 2020, after 2.5 years of seismic quiescence in South Baikal, the first hydroisotope signs of deformation instability appeared at the Kultuk test site at the end of March. A tangible seismic shock occurred in the water area of the lake three months later, on July 6, and on the night of September 21–22, near the village. A stronger earthquake followed soon. Online observations of deformations in an adit at the test site revealed the last, six-day phase of preparation of the Bystrinskoe earthquake. It triggered seismic instability in the entire South Baikal Depression and reverberated with a new strong earthquake on December 9 in the delta of the Selenga River, near the Proval Bay.
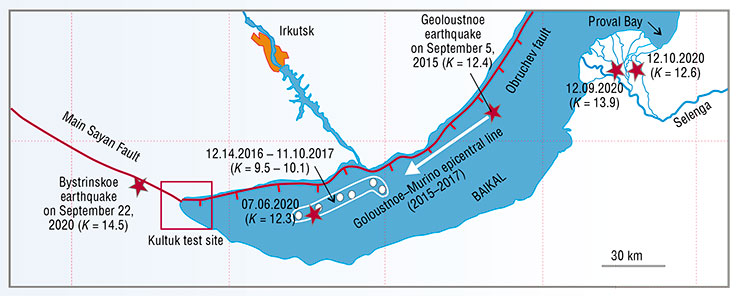
Today, predicting strong earthquakes in South Baikal in the medium (months) and short term (days or hours) is becoming a reality because of the efforts of researchers from the Institute of the Earth’s Crust: V. V. Ruzhich, S. A. Bornyakov, K. Zh. Seminsky, A. M. Ilyasova, and others, as well as E. P. Chebykin from the Limnological Institute.
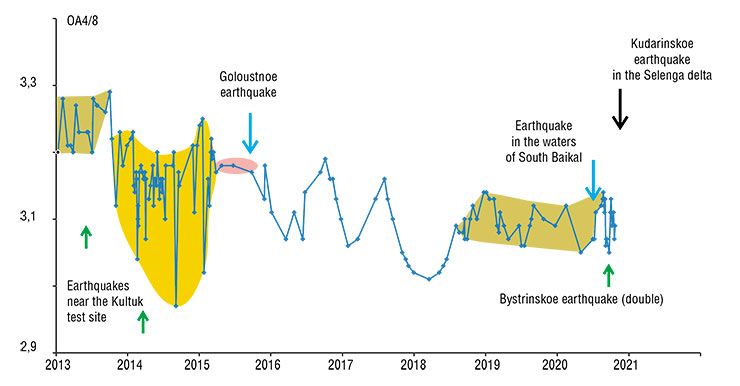
Monitoring of the Cherdyntsev–Chalov effect in groundwater is an integral part of the comprehensive observations of seismogenic deformations in active faults, which are organized within a large-scale project of the Russian Ministry of Education and Science “Fundamental Principles, Methods, and Technologies for Digital Monitoring and Prediction of the Ecological Situation in the Natural Territory of Lake Baikal.” Specific predictive conclusions depend on the frequency of sampling at the monitoring stations and on timely measurements of uranium concentrations and AR4/8 in groundwater samples.

References
Chebykin E. P., Rasskasov S. V., Vodneva E. N., et al. First results of 234U/238U monitoring in water from active faults on the western coast of South Baikal // Doklady Earth Sciences. 2015. V. 460. N 2. P. 142–145.
Rasskazov S. V., Chebykin E. P., Ilyasova A. M., et al. Creating the Kultuk polygon for earthquake prediction: variations of (234U/238U) and 87Sr/86Sr in groundwater from active faults at the western shore of Lake Baikal // Geodynamics & Tectonophysics. 2015. V. 6. N 4. P. 519–554.
Rasskazov S., Ilyasova A., Bornyakov S. et al. Responses of a 234U/238U activity ratio in groundwater to earthquakes of the South Baikal basin, Siberia // Frontiers Earth Sci. 2020. V. 14. P. 711–737.
Rasskazov S. V., Ilyasova A. M., Chuvashova I. S., et al. The 234U/238U variations in groundwater from the Mondy area in response to earthquakes at the termination of the Tunka Valley in the Baikal Rift System // Geodynamics & Tectonophysics. 2018. V. 9. N 4. P. 1217–1234.
Rasskazov S. V., Ilyasova A. M., Chuvashova I. S., et al. Hydrogeochemical zoning of uranium isotopes (234U/238U) in the Southern Siberian paleocontinent: the role of the South Baikal reservoir in the groundwater formation // Geodynamics & Tectonophysics. 2020. V. 11. N 3. P. 632-650.
Seminsky K. Zh., Bornyakov S. A., Dobrynina A. A., et al. The Bystrinskoe earthquake in the Southern Baikal region (21 September, 2020, MW = 5.4): main parameters, precursors, and accompanying effects // Russian Geology and Geophysics. 2021. V. 62. N 5. P. 589–603.


















