Siberian Center for Genetic Resources: Three Years Later
A year ago, the infrastructure of the Siberian Branch of the Russian Academy of Sciences (SB RAS) gained a facility unique for Russia, an SPF vivarium meeting all the international standards, which was organized with the Institute of Cytology and Genetics.
“Science First Hand” has already informed its readers about the construction of this technically most intricate facility intended for keeping almost “sterile” laboratory animals used in experimental research. The new shared access center has become the basic experimental facility for multidisciplinary research of scientists from many institutes with the Siberian Branch of the Russian Academy of Sciences
The principle to treat a patient rather than a disease, stated by Hippocrates as early as the 4th century BC, is becoming ever more important given extensive investments in pharmacology. Currently, the treatment efficacy even with the most state-of-the-art drugs does not exceed 60 %, while negative side effects are observed in approximately one-third of the patients (Kukes et al., 2008). Hence the centuries-long history of search for the approaches to personalized medicine based not only on the expertise and intuition of the physician, but also on an objective evaluation of individual features of a patient now gets a second breath.
The reason for optimism is the list of scientific achievements headed by sequencing of the complete human genome, completed at the turn of the second millennium. There are no doubts that due to the technological breakthrough the cost of reading personal genomes will be soon covered by a common medical insurance. Concurrently with advances in the genetic analysis technologies, information about the effects of individual genes on human predisposition to various diseases or, on the contrary, to achieving brilliant success in intellectual, creative, or sport activities is accumulated.
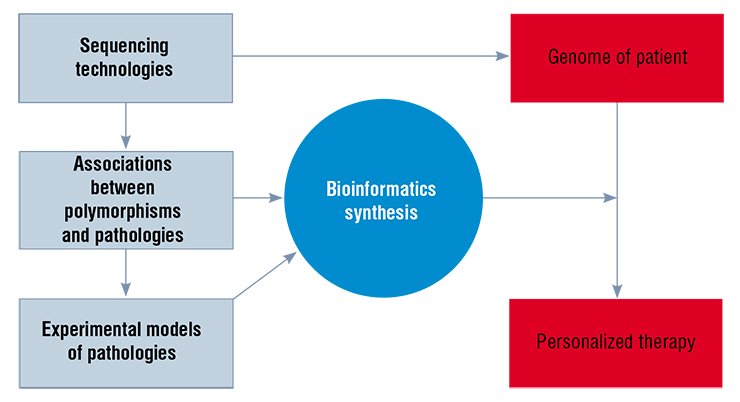
According to expert estimates, the number of such genetic variations (polymorphisms) exceeds two million (Bloom, 2001). State-of-the-art information and computational technologies allow individual genes or their complexes specifying particular individual predispositions to be detected among this multitude.
However, would this be sufficient for developing the optimal strategies for personalized disease prevention and therapy or for establishing personal educational “itineraries”? The fact is that the human body, as well as that of the most closely related mammalian species, is extremely complex. Therefore, an insight into the mechanisms underlying implementation of the genetic programs, including those leading to diseases, requires experimental studies of the animals closest to humans in their genetic and general biological characteristics.
Fellows in illness
To model genetic predispositions to various diseases, laboratory animals are traditionally selected for the presence of the symptoms characteristic of a target pathology. Thus, the rat and mouse strains displaying arterial hypertension, premature aging, and several behavioral deviations from the “norm” have been bred at the Institute of Cytology and Genetics SB RAS. Such organisms are precious material for an insight into pathogenetic processes and search for new disease prevention tools and remedies.
In addition, the carriers of spontaneous or experimentally induced mutations leading to various abnormalities in morphology, metabolism, or physiological processes characteristic of some human diseases have been discovered among laboratory animals.
However, a real golden age in creating experimental animals with genetic predisposition to diseases came in the postgenomic era, i.e., at the beginning of the 21st century. Currently, genetic engineering methods make it possible to directly switch off (knock out) individual genes or to produce the animals with variants of the genes associated with human diseases inserted into their genomes.
The study of rheumatoid arthritis conducted at the University of Tokyo provides an example of a successful attempt in this field (Iwakura et al., 2008). Many of the rheumatoid arthritis patients in Japan are carriers of the retrovirus HTLV-I. To clarify its role in the corresponding pathogenesis, researchers inserted this retrovirus into the mouse genome; the corresponding mice, from a certain age, started to show typical symptoms of rheumatoid arthritis. Another mouse strain was obtained for comparative analysis: in these mice, a knockout of one gene responsible for producing the antiinflammatory regulator IL-1 Ra caused the predisposition for arthritis. Then all the key genes of inflammatory responses that determine the disease development were knocked out in a stepwise manner in each of the strains.
The genetically engineered mouse strains eventually allowed the most adequate molecular targets for pharmacological impact to be detected. These targets appeared to be different for arthritis determined by different causes. It has become clear why drugs efficient for the majority of Europeans are useless for most Japanese patients. This is an example of a scientifically grounded approach to the development of personalized medical technologies when applied to a particular disease.
The studies of rheumatoid arthritis are only one example of specific ethnic features in the development of the diseases that can be most similar in their symptoms. Taking into account these specific population features plays an important role in the establishment of personalized medicine. This also makes us abandon the illusory hopes for solving our own problems only at the expense of knowledge import. Therefore, expansion of research in the field of experimental modeling of various pathologies in the countries committed to national health protection is supported not only (and not as much) by the pharmaceutical business but rather by federal funding. It should be underlined that the ethnic aspect is of paramount importance for such a multinational country as Russia.
In mouse coding
To clarify only two variants of pathogenesis in the case of rheumatoid arthritis it was necessary to breed eight mouse strains with specified genetic characteristics. However, the number of diseases, to say nothing about their individual variants, is considerably larger.
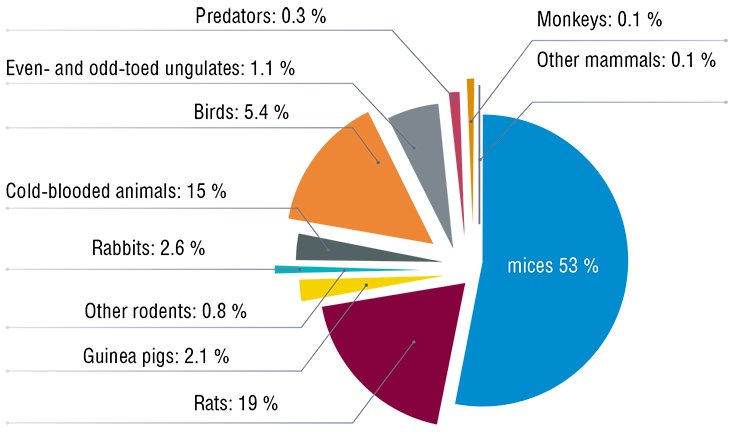
Actually, so many patients, so many variants. It is unfeasible to provide each of them with an experimental model; however, it is necessary to grasp the major principles underlying the development of genetically determined diseases. This fact implies an explosive growth in the stock of experimental animals, observed in all developed countries.
In addition to the simulation of pathologies, creation of collections of experimental animals whose genomes are closely related to the human genome is among the key conditions for advancing from the determination of “sequences of letters” in the human genome to a meaningful interpretation of this most intricate text.
The whole mouse genome was sequenced directly after the human genome sequencing. Almost immediately, it became clear that the mere knowledge of nucleotide sequences does not allow the effects at the level of an integral organism to be predicted (Schofield, 2008). The simplest approach to answering the question on the role of a particular gene in the development of individual properties of an organism is in studying the phenotypic* effects caused by a knockout of this gene. For this purpose, it is necessary to have collections of knockout strains whose number should be close to the total number of all genes.
A large scale of this problem does not intimidate modern scientists. An example of their gallantry is the Knockout Mouse Project (KOMP), initiated by the National Institutes of Health of the United States in 2005. The goal of this project is to create a collection of genotypes each of which carries one knocked-out gene, using a “popular” mouse laboratory strain, C57Bl. Three additional research teams shortly joined this project, founding the International Knockout Mouse Consortium (IKMC).
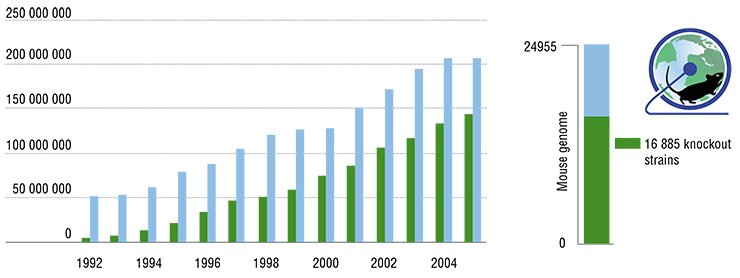
Currently, their joint efforts allowed them to produce over 16 000 embryonic stem cell lines with knocked-out coding genes (note for comparison that the complete mouse genome contains about 25 000 genes). So far, these cell lines have been used to create over a thousand and a half mice with individual knocked-out genes.
In addition to gene knockout, genetic engineering approaches allow animals with controllable gene expression (activation) to be obtained. This control is available at any moment of an individual life by administering pharmacological preparations. Moreover, mouse strains with gene activation coupled to the synthesis of fluorescent proteins are produced, which provides for direct observation of how genes work. This allows researchers to monitor, literally “with their own eyes” – with the help of optical probes or directly through the skin – the genetic processes underlying embryonic development as well as various behavioral, physiological, or pathogenic processes.
Finally, transgenesis has been used to produce the so-called humanized mice, some genes of which are replaced with human genes. These animals are used to assess the effect of a drug on molecular targets, which in this case are fully “human.”
In the case of the genes coding for immunoglobulins, such substitution allows the mouse immune system to be used for producing therapeutic human monoclonal antibodies (Jakobovits et al., 2007). The first such immunoglobulins that passed clinical trials were the antibodies to interleukin-8. These antibodies were used to inhibit immune processes in the patients with relapsing psoriasis. It is of fundamental importance that no patient displayed an allergic reaction. Currently, over 10 variants of the human immunoglobulins produced using mice and intended for use as antibodies to various growth factors of malignant tumors are undergoing preclinical trials.
From gene to organism
Thus, mouse genotype exists as three hypostases: an object of research, a research tool, and a production facility in the biotechnological cycle. Therefore, it is not at all surprising that, according to the prediction in Nature (2004), by the year 2025 the number of mouse genetic strains will increase to 300 000.
The current advance in technology, permitting a targeted impact on individual genes and estimation of their activity, has created conditions for efficient solution of a most intriguing problem in systems biology, namely, construction of the so-called functional trajectories from genes to adaptive or pathological responses of an integrated organism.
Along with genomics and proteomics studies, a key part in solving this problem is assigned to the analysis of morphological, physiological, and behavioral processes, united within the concept of phenotyping. This field has recently gained considerable methodological support owing to the advances in the creation of the above-mentioned systems for intravital recording of fluorescent agents, including those formed when individual genes are activated; computerization of the systems for observation of animal behavior; and design of miniature implantable detectors, including a miniature video camera implantable into the brain.
Magnetic resonance imaging (MRI) scanners stand alone in the toolkit for intravital monitoring; they make it possible to control the morphological and physiological changes in various body systems, estimate the uptake and excretion of physiologically active substances, and even measure the contents of various products of metabolism (metabolites) at any site of a living animal.
An unrestrained increase in the number of genetic strains modeling various properties of the body, including hereditary diseases, development of intravital examination technologies, and the need to standardize the conditions for animal manipulations have led to the establishment of special objects in the science infrastructure – centers for genetic resources of laboratory animals.
Mouse Resources
In developed countries, specialized centers for genetic resources, intended for supplying basic and applied research with laboratory animals, have a national status. Their basic component is vivariums, where SPF (specific pathogen free) animals are kept under sterile and stringently standardized conditions in terms of the diet, water supply, and microclimate, which is of paramount importance for the reproducibility of experimental results obtained in different laboratories.
Standardization grows in importance when problems in postgenomic biology are addressed, because phenotyping of numerous genotypes produced by genetic engineering methods requires joint efforts of research laboratories from different countries. In addition, retention of an SPF status is the only possible way for immunodeficient genotypes to survive and reproduce.
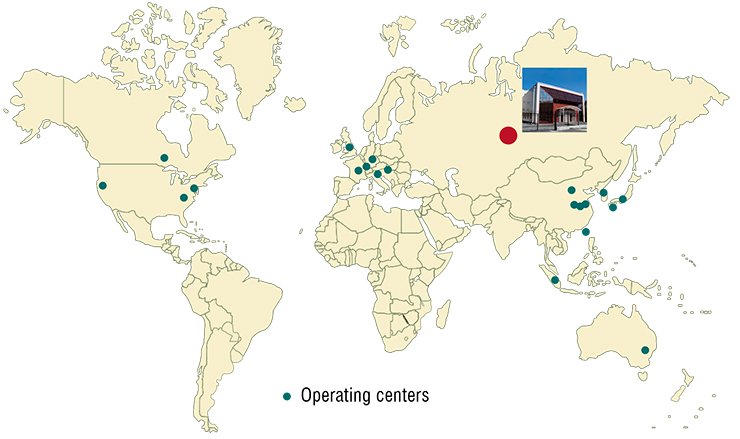
At the international level, activities of the centers for animal resources are coordinated by several institutions, such as the Federation of International Mouse Resources (FIMRe) and Asian Mouse Mutagenesis and Resource Association (AMMRA), uniting over 25 centers in different countries. The total genetic diversity compiled in these centers approaches 30 000 mouse genotypes.

The centers exchange strains in two ways, namely, shipping traditional breeding stocks or sending frozen embryos in special containers filled with liquid nitrogen. The second way is necessary because traditional breeding, even of the already existing genetic strains, is very expensive. As a consequence, modern centers for genetic resources have their own cryobanks and laboratories for reproductive technologies, where the overall operation cycle is performed: production of embryos at early developmental stages (two to eight cells); their freezing and storage at the liquid nitrogen temperature; their thawing and transplantation to future mothers for producing offspring and subsequent breeding.
What is the situation in Russia? Progress made in Russia’s accession to the WTO entails several issues, including those related to the experiments with laboratory animals. In particular, all the laboratory animal facilities in this country should be reorganized to meet the international standards.
So far, in addition to the animal breeding facility and laboratory for biological trials with the Pushchino Branch of the Institute of Bioorganic Chemistry, Russian Academy of Sciences, which have an international accreditation, several other SPF animal breeding facilities have been constructed and put in operation. They are the vivariums of the State Research Center of Applied Microbiology (Obolensk, Moscow oblast); Institute of Physiologically Active Substances, Russian Academy of Sciences (Chernogolovka, Moscow oblast); Gamaleya Institute of Epidemiology and Microbiology, Russian Academy of Medical Sciences; and Moscow State University. The main goal of these institutions is to conduct preclinical trials of drugs using widespread laboratory animal strains, whose main source is the Pushchino animal breeding facility, keeping eight mouse and four rat strains. Recently, the first transgenic and knockout animals have appeared in this facility.
However, the Russian Federation still lacks a specialized center for genetic collections of laboratory animals that would be associated with international federations (FIMRe or AMMRA) and thus gain free access to the overall world abundance of model organisms. All this hinders greatly both basic and innovation research in the most advanced fields of knowledge. This is why foundation of a state-of-the-art center for genetic resources can be regarded as a most topical challenge and a must for the further development of competitive Russian science.
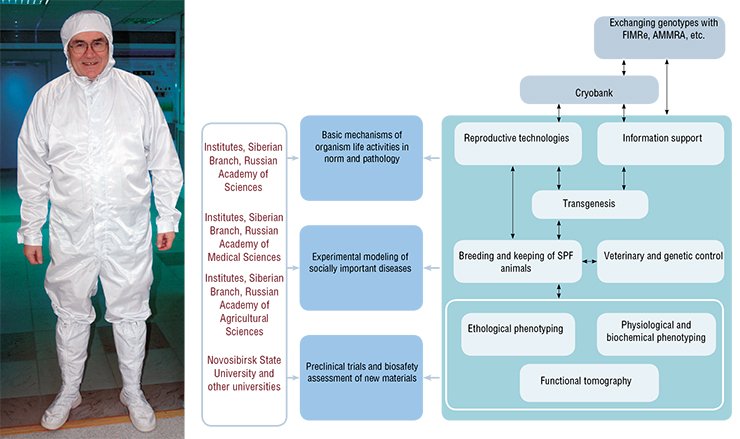
Today, the role of such a center can be claimed by the inter-institutional shared access center SPF Vivarium with the Institute of Cytology and Genetics SB RAS. The center, whose construction was funded by the Ministry of Economic Development, was put in operation in 2009.
Sciences arm in arm
The construction and technological development concept of the first center in Siberia and Russian Far East meeting all the international requirements on maintaining experimental animals is based on the principle of multifunctionality. This makes it possible to combine basic research, associated with phenotypic studies of genomes, with applied work in the fields of biomedicine, pharmacology, and nanobiosafety. The center can provide for cryopreservation of tens of thousands of genotypes, maintenance of up to one hundred nuclear stocks of mice and rats, and studies meeting all the GLP (good laboratory practice) requirements under super sterile conditions of the containment zone with an area of 1400 m2. This resource and research facility is unique for Russia.
Today, we have already acquired some experience of using it and obtained the first results.
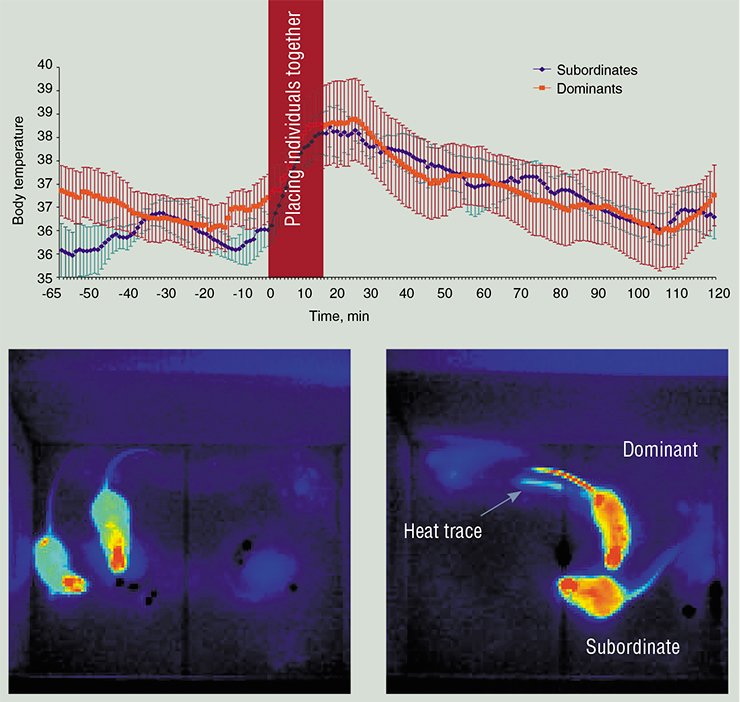
TAIL AS THE WINDOW TO THE SOUL Joseph Barcroft, an outstanding British physiologist, was amazed by how pronounced were the specific individual features in the regulation of vascular tone in the hour of danger: London residents looking for shelter from air raids in the Tube either blushed or paled. You cannot observe such external manifestations of emotions in laboratory animals – however, emotions show themselves if the tail skin temperature is recorded.Recording the internal temperature and heat flows from the body surface enriches behavioral testing, used in neurophysiological and pharmacological studies, with the information about vascular responses of an individual to emotionally significant stimuli. Another field of application for these methods is monitoring of tumor development or inflammation foci, for example, when testing new cosmetic creams or aerosols.
However, to make devices for recoding temperature responses a common tool in behavioral and biomedical studies, it is necessary to adapt them to experimental conditions. Only while performing biological tests, it is possible to elaborate software designed for automated processing of recorded signals and their conversion into a terminology clear for biologists.
Currently, the SPF Vivarium has all the necessary conditions for experimental adjustment of the equipment and methods developed at institutes of the Siberian Branch of the Russian Academy of Sciences, which can considerably expand the field of application for the devices initially intended for solving specific problems
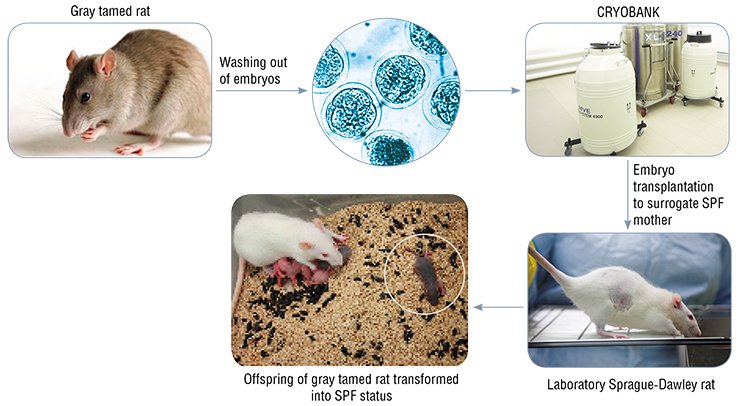
Since the first year of the SPF vivarium operation, the first mouse and rat genetic strains, including two knockout genotypes, have been living there. By the end of 2010, a unique strain of “tamed” rats, bred at the Institute of Cytology and Genetics from the rats’ wild ancestors, was successfully transformed into an SPF status. For this purpose, the embryos were transplanted to sterile surrogate mothers. The success of this work demonstrated that the cryoarchiving complex was ready to collaborate with the world centers for genetic resources.
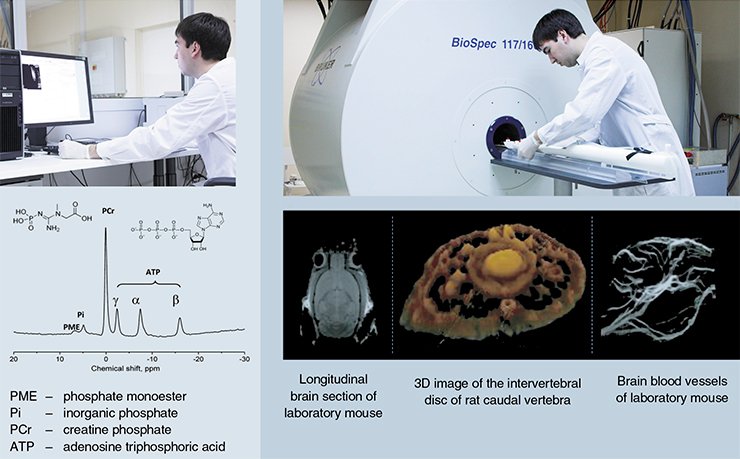
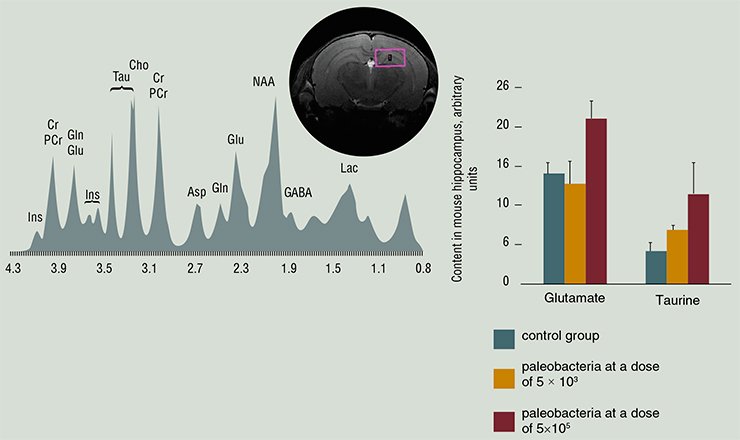
After a tryout of the unique MRI scanner, an inter-institutional tomography sector was organized at the SPF Vivarium facilities, in collaboration with the International Tomography Center. During the first year of the SPF Vivarium operation, over 300 different laboratory animals were examined there. The result of these studies were detailed morphological descriptions of the brain structures in the mouse and rat genetic strains simulating various socially important pathologies, such as hypertension, premature aging, and mental disorders. The availability of magnetic resonance tomography (MRT) for animals allowed researchers from the International Tomography Center and other physical and chemical institutes to accelerate testing of new technologies, including the testing of original contrasting agents.

Another area in which the efficiency of collaboration between professional physicists, chemists, biologists, and mathematicians distinctly manifested itself was rapid mastering of the most advanced tomography methods. In particular, the first data on a spatial distribution of the mouse brain regions responding to socially significant stimuli were obtained using functional tomography. Intravital NMR spectroscopy initiated the study of the metabolomic profiles of the brain in different genetic strains, including the testing of therapeutic and preventive drugs.
NO HAND HOLD! Genomics, transcriptomics, proteomics, metabolomics, and phenomics are neologisms of modern biology that mark the milestones on the way where genetic “plans” are put into phenotypic practice. Metabolomics – next to last but not least in this list – deals with studies of metabolism, that is, the totality of all the intermediate and final metabolic products in the cell, tissue, organ, or whole body. Quantitative determination of individual compounds and whole compositions characterizes metabolic processes in living structures as well as reflects the state of their “habitat.”NMR spectroscopy makes it possible to determine the content of metabolites at any site of the body without any damage to the organism itself. There are only two limitations, namely, the corresponding site should be motionless for several minutes (in the case of regular breathing vibration, a virtual fixation of the analyzed area is possible) and its size should be at least 8 mm3. The brain is the most advantageous and interesting object.
The proton and phosphorus NMR spectroscopy has been mastered at the Interinstitutional Sector of Tomography of Laboratory Animals. It took less than half year to perform a number of studies showing good prospects of intravital spectroscopy for studying the neurochemical foundations of animal behavior.
Another source of noninvasive metabolomic information is low molecular weight compounds contained in the body excretions. These compounds are efficiently detected with the help of chromatography and chromato-mass spectrometry methods; however, a qualitative separation of complex chemical mixtures is usually a time-consuming process.
Creation of portative high-throughput polycapillary chromatographs allowed the gap between the biological studies of odors and determination of their chemical nature to be overcome. An example of such a device is the EKhO-V-FID chromatograph, designed at the Institute of Petroleum Geology and Geophysics SB RAS. This chromatograph has provided for analyzing the gaseous phase of urine samples of control and antigen-stimulated male mice and allowed the chromatographic peak responding to activation of the immune system to be identified. Concurrently, the compounds whose level correlated with the reproductive status of males were identified in these samples.
For the moment, noninvasive metabolomics still plays a modest role among the methodical approaches used for describing new genotypes of laboratory animals. However, its significance will increase with the development of analytical technologies and decrease in their cost. In addition, the diagnostically significant patterns thus discovered can be used directly in medical and veterinary diagnosing
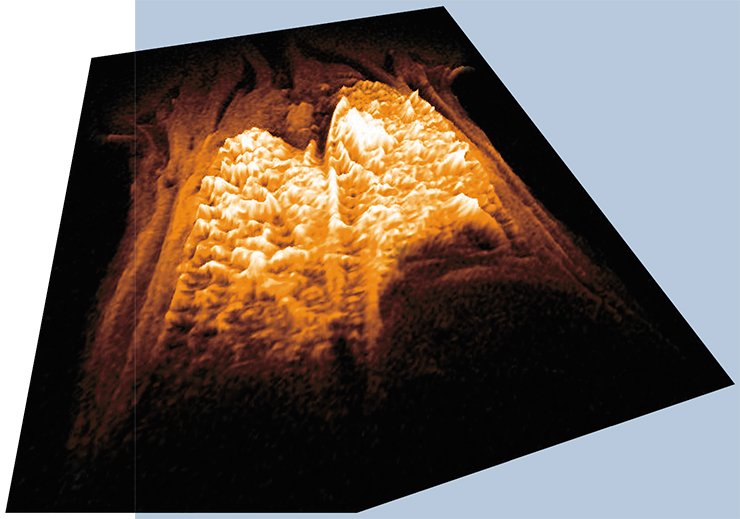
The first important scientific task jointly fulfilled by researchers from seven Novosibirsk academic institutes at the facilities of the SPF Vivarium was an assessment of nanomaterials biosafety. For the first time in Russia, these studies have been performed in full accordance with the international GLP standard. This work had required certain prearrangements, namely, a toolkit for measuring the sizes and shapes of nanomaterials as well as a device for exposing animals to aerosols containing silicon dioxide nanoparticles (Tarkosil 25).
The MRT has been also used to obtain 3D images of the lungs allowing for a quantitative estimation of the distribution of nanoparticles in them. A study of the integrated immunophysiological response of mice to the nanopowder inserted in their upper airway has demonstrated that the pulmonary immune response is observed only when the particles are smaller than 100 nm and only in the genetic strain with prevalence of the so-called humoral type of immune response. The discovered genotype-dependent effects of nanoaerosols may be important in developing the criteria for occupational selection of the staff engaged in nanomaterials manufacture.

Sequencing of the human genome as well as of the genomes of closely related animals has revolutionized the entire network of research into the genetic diversity of mammals. Modern biotechnologies have made laboratory animals the most important resource of postgenomic biology, which is well illustrated by an exponential growth in the number of research papers on the studies involving transgenic animals. In particular, the number of papers with “transgenic mouse” as a key word has increased by more than 30 000 over the last 2.5 years!
However, the human nose in its structure is among the simplest noses. The mouse nose is full of diverse olfactory receptors. According to layer-by-layer NMR images of the nasal passages of laboratory mice, their intricate geometry suggests a considerably more complex structure of the air flows as compared with those of humans.
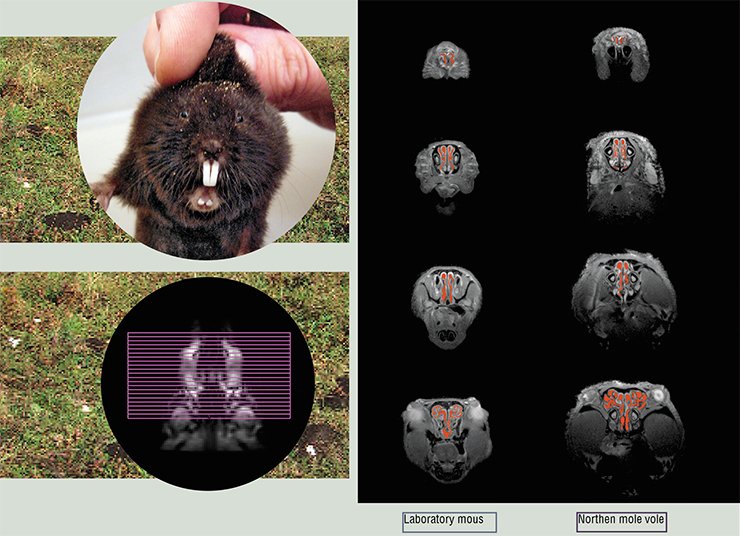
The nose of the northern mole vole, a small rodent making numerous underground tunnels in its search for food (plant roots) has a much more complex structure. The energy expenditure during digging increases two- to threefold; respiration intensity and probability of dust entering the lungs increase correspondingly. Tomography of the northern mole vole nose has demonstrated two interesting specific features: the area of the cross-section through the nasal cavity in its maximum extension is 20-fold that of its entrance, which is vastly larger than in mice; and the lower part of the nasal passages contain fluid, which is absent in mice and other land mammals.
A complex shape of the nasal passages and the presence of fluid allow the nose of the northern mole vole to be compared to a vacuum cleaner with a water filter. The efficiency of such protection will be demonstrated by aerodynamic models as well as by keeping these animals in the atmosphere with dust particles. A more comprehensive study of this species, adapted to live in a dusty environment, can contribute to the elaboration of new protection tools for the human respiratory system against the impact of particles of various sizes. Researchers from four institutes of the Siberian Branch now tackle this problem. It is encouraging that not only unique scientific technologies are the reason for integration, but also unique animals living in Siberia
The need to accumulate specific knowledge about the implementation of genetic information is caused not only by an incredible complexity of living beings, but also by the absence of a “typical design” within each biological species, humans included. This is why an ever increasing number of manifold living genetic “models” are brought into the biomedical research focused on the search for the methods that treat “patients rather than diseases.”
One of the stimuli for creating these models is rapid accumulation of information about “gene-pathology” associations. The possibility to study an animal whose genotype meets the need of researchers sets a new vector in biological research, which requires integration of multidisciplinary research efforts and multinational financial investments.
The editorial titled “Mouse Megascience,” published in Nature (June 03, 2010), compared the studies into genetic diversity of laboratory animals, in terms of their scale and significance, to the Large Hadron Collider megaproject. Biologists are called to unite to put educational pressure on politicians with a view to redirecting financial flows to the socially significant scientific activity investigating the basic foundations of human health.
To ensure adequate interaction with the world science in this field, Russia needs at least one center for genetic resources that would meet the international requirements, and such a center is now being established with the Siberian Branch of the Russian Academy of Sciences, at the facilities of the SPF Vivarium (Institute of Cytology and Genetics). Such a center is of interest for many academic institutes as well as for classical, medical, and agricultural universities and for the reviving pharmaceutical industry.
Since gene frequencies associated with a disease and the pattern of these associations itself may have certain specific ethnical features, creating conditions for developing scientific foundations of personalized medicine should be a national priority. First and foremost, this should refer to the centers for genetic resources, since they are an integral component of the science and technology called to meet one of the main challenges any state faces – maintenance of the nation’s health.
References
Moshkin M. P. POSTGENOMNAJA JeRA ili Zachem nuzhny 300 tysjach linij myshej // NAUKA iz pervyh ruk, 2008. № 4 (22). S. 16—53
* Phenotype is the totality of all the traits and properties of an organism formed during its individual development













