Tissue Engineering: A 3D Angioplasty
Adverse ecological conditions, sedentary lifestyle, imbalanced diet, and stresses—all these factors contribute to an increase in the number of vascular diseases. Healing these illnesses frequently requires transplantation, a surgical intervention involving the replacement of tissues or whole organs. However, current medicine is sometimes unable to help such patients, first and foremost, because of deficiency in donor material, tissue compatibility problem, and absence of the suitable artificial prostheses. State-of-the-art molecular biological approaches and cell engineering have made it possible to closely approach the construction of artificial prostheses, including the polymeric blood vessels, which almost do not differ from their natural prototypes in their properties
Organ and tissue transplantation is one of the topical problems in current medicine including in no small part vascular surgery. Today, the world demand for small-diameter vessels for aortocoronary bypass is no less than 450,000 annually, to say nothing of the prostheses for restoring the blood circulation in lower limbs, aortorenal bypass surgery, and graft of other small-diameter arteries.
Transplantation requires donor veins, animal vascular xenografts, and, finally, grafts of synthetic polymeric materials. Note that the natural implants have several essential disadvantages, including the problems stemming from immunological compatibility, change in the implant size with time, possible misfit in size of the implant and replaced organ, as well as manifold issues associated with obtaining and storage of the donor material. The artificial implants are free of some disadvantages and allow the blood circulation to be restored even when the vessel damage is considerable. On the other hand, the use of artificial implants is fraught with the development of inflammatory foci and stenosis (a narrowing or stricture of a vessel.)
The currently available artificial vascular implants differ from their natural prototypes in mechanical properties; in addition, the former poorly integrate into the patient’s tissues and a normal cell layer, endothelium, fails to develop on their surface. Such implants are inappropriate for repairing small-diameter (less than 6 mm) vessels. Despite a considerable advance achieved in the last 50 years in the creation of materials for vascular prostheses , such as Dacron and polytetrafluoroethylene, the possibilities of these materials have been so far almost exhausted.
However, we can see “light at the end of the tunnel:” an intensive development of biochemical, molecular biological, and cell engineering methods has made it possible to closely approach the construction of almost perfect prostheses of various organs. In particular, artificial trachea and esophagus, comprising several tissue types and almost identical to the corresponding organs were designed (Curcio et al., 2010; Liang et al., 2010). The production technology for such prostheses includes creation of a 3D scaffold (or matrix) of the target organ, which is then “populated” with stem cells or the corresponding differentiated cells.

The scaffolds may be either biological or synthetic. In the former case, a donor organ is used, which is freed from all cells so that only the framework of connective tissue proteins is left; the purpose is to maximally reduce the risk of immunological incompatibility of the prosthesis and its future host. Then, the necessary tissues are “grown” on this scaffold from the cells of the future recipient (Badylak et al., 2012). However, such implants retain certain shortcomings characteristic of donor organs, including the risk (although small) of immunological incompatibility.
An alternative is the entirely artificial scaffold structures, safe and displaying unique properties owing to novel composite materials and the methods used for their production.
Electrospinning: an ocean of possibilities
Currently, there are several methods for producing 3D scaffolds of polymeric materials, such as molding (casting or pressing), phase separation, and 3D printing.
Electrospinning holds a special position among these methods, being a unique technology allowing polymeric fibers with a thickness of several nanometers to several microns to be produced from polymer solutions, which is unfeasible by any other methods for fabrication of nonwoven materials. The structures formed of such fibers have a vast surface area and high specific strength, while different methods of fiber packing make it possible to obtain anisotropic materials of manifold shapes with any degree of porosity and density.
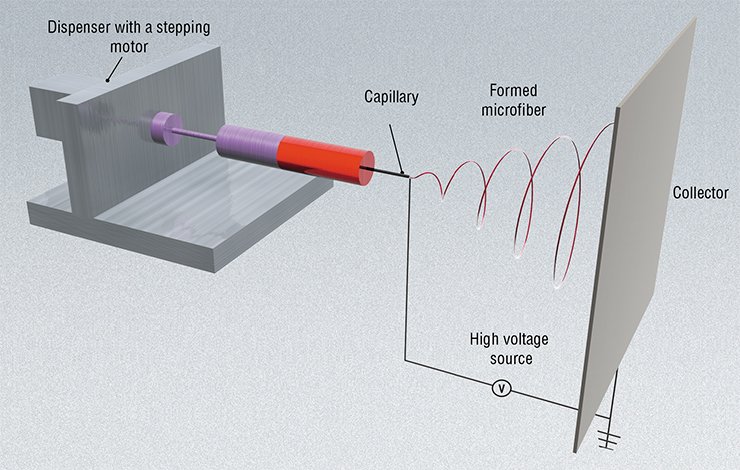
ANTAGONISM OF FORCESThe principle of the method is that drops of water (or a polymer solution) flow out through a dosing capillary from a reservoir connected to high voltage; as a result, electric charge accumulates on the surface of a drop. The drop is formed in an electrostatic field, the vector of which is directed against the surface tension force of liquid. At considerably high values of the electric field strength, the drop starts to elongate to eventually form the so-called Taylor cone. If the value of the charge applied on the drop surface exceeds twofold the applied Rayleigh limit, when the Coulomb repulsion of charges on the surface is equal to the surface tension force, the Taylor cone is deformed and falls into many droplets.
After the evaporation of solvent, the drops of the solution form particles. Unlike the particles produced by routine mechanical methods, such particles are smaller (micro- and nanoscales), more homogeneous, and almost do not aggregate, while their motion and distribution is easily controllable by varying the strength of the applied electrostatic field. Changing the field strength and polymer concentration in solution, it is possible to produce particles of different diameters and by supplementing solution with additional component, to modify the properties of particles.
During electrospinning, the Taylor cone does not fall into particles but rather forms a jet of solution, which is directed towards the collector electrode by the applied electrostatic field, becoming longer and thinner on its way. Over the time required for the solution jets to reach the surface of collector, the solvent evaporates, and the ready polymer fibers are deposited on the collector
This technology utilizes electrohydrodynamic spraying of liquids (electrospray), when a liquid flowing to the feeding capillary is nebulized into small drops under the impact of applied electric field. When a polymer solution is used, this may give rise not only to drops, but also to polymeric fibers (this is electrospinning per se).

The way that the process will follow as well as the diameter and structure of the resulting fibers depends on many factors, such as the viscosity and conductance of the used solution, properties of the solvent, and field intensity. In particular, the degree of fiber uniformity and the number of drop-like bulges are determined by the concentration of the polymer solution (Fong, 2009). The surface tension of the solution also influences the uniformity of fibers, and since this tension is mainly determined by the properties of the solvent, it is the major contributor to the fiber ultrastructure. Although the theory of this process has been described in many papers, the actual conditions for fabricating materials with specified properties are usually selected in an empirical manner.
CLASSIFIED FOR 50 YEARSThis discovery was forgotten for almost a century and a half; however, electrodynamic spraying was widely used in the 20th century for engineering purposes, in particular, generation of monodisperse aerosols, atomizing of dielectric and cryogenic fluids, targeted deposition of pesticides and polyfunctional coatings, and jet printing.
As for fibers, the first patents on fiber production of a solution jet injected into a space with a strong electric field were issued in the United States as long ago as 1902. In the 1930s—1940s, the patents on the fabrication of fibers of artificial silk, elastic gum, cellulose esters, and so on were also issued. Nonetheless, the commercial fiber production by this technique was absent abroad until the 1990s because of a poor fiber quality and low competitive ability (Filatov et al., 2008).
A real breakthrough in this field was made in the late 1930s, when N.D. Rozenblyum and I.V. Petryanov-Sokolov, young researches from the Laboratory of Aerosols with the Karpov Physiochemical Institute (Moscow), attempting to electrospray monodisperse aerosol particles, encountered a competing mode when fibers were generated. In 1938, they produced the first mats of microfibers, which were proposed as an efficient smoke filter.
Commercial production of the Petryanov filter was organized in a year to be used as smoke filters in gas masks. A paramount importance of fiber application resulted in that the corresponding technology was classified for 50 years (Filatov et al., 2008).
Initially, the filters were made of cellulose acetate and then other polymers were also used. The overall annual production of electrospun materials in Russia reached 20 million m2!
Currently, because of the interest of global industry in nanomaterials and nanotechnologies, electrospray and electrospinning are in the focus of attention of scientific community: over 200 research institutions worldwide are involved in designing new fiber materials
Electrospinning makes it possible to create an infinite set of structures differing in their composition and physical properties. In the easiest case, a fiber made of one solution is deposited on a flat collector electrode, which gives an unarranged 3D matrix. However, it is also possible to fabricate various ordered structures, for example, honeycomb-like ones, using controlled collectors, such as a revolving drum or a disc. In the latter case, an ordered matrix in the form of a flat sheet, fiber, or tube is produced. By varying the rate of deposition, we can vary the density, porosity, and stability of matrices.

In a more complex case, the matrix is produced of two or more types of fibers using the solutions of different polymers, either concurrently or in turn. As a result, either a uniform or a layered matrix is formed. This approach allows for the fabrication of more porous or more stable matrices as well as the matrices able to change their properties with time. One of the interesting variants of this technology is coaxial electrospinning, which gives coaxial fibers, that is, the structures with an envelope and core. In such fibers, the inner layer is protected from environmental impacts, making them promising tools for drug delivery in a prolonged manner.
An ideal substitute
Solutions of both synthetic polymers and polymers of biological origin are appropriate for electrospinning. Animal fiber proteins (collagen, elastin, and fibrin) as well as polysaccharides of plant (for example, cellulose and pectin) and animal (chitosan, hyaluronic acid, and chondroitin sulfates) origins are used as natural biopolymers. Electrospinning can produce fiber structures not only of polymers, but also of lipids. For example, the fibers of lecithin, known for its ability to form micellar and fiber structures, have been already produced.
Of synthetic polymers, nylon, polycaprolactone, polylactide-co-glycolide, and their copolymers are mainly used for this purpose. The items from nylon degrade slowly in the body but lose elasticity with time. The items of more stable polycaprolactone retain stability for 1 to 3 years depending on the degree of polymerization. The items of biodegradable polylactide-co-glycolide have different degradation rates and are preserved in the body for several months. The last polymer is optimal for the implants intended for resolving in the body; it is frequently used for manufacturing suture material (for example, Vicryl).

Various low- and high-molecular-weight substances, such as antibiotics, cytostatics, cell proliferation stimulators, and other drugs may be added to the polymer solution when biological matrices are produced. By influencing the cell environment of the graft, such items will “control” the rearrangement and regeneration of the surrounding tissue and stimulate or, on the contrary, suppress cell growth as well as inhibit inflammation and infection.
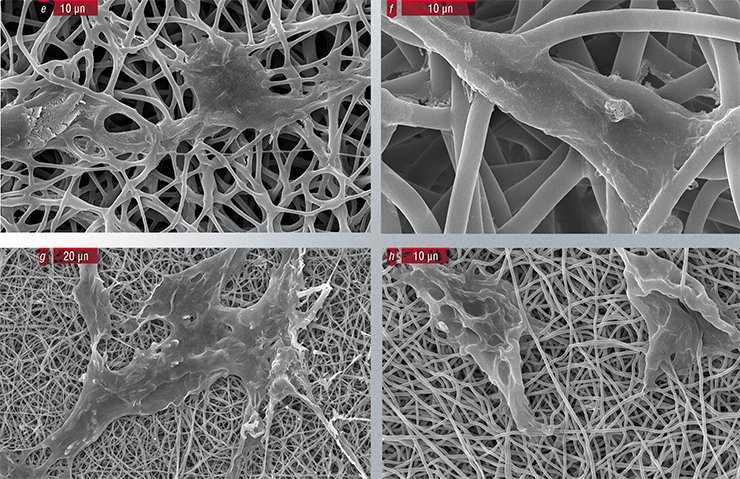
It has been shown that the polymeric matrices produced by electrospinning can be successfully used for the regeneration of various tissue types. For example, such matrices efficiently supported the growth of the cartilage, connective, muscle, and even nervous tissues in cell cultures. Moreover, experiments with animals have demonstrated that they display a high biological compatibility, do not cause inflammation, and are efficiently substituted by a normal tissue (Agarwa, 2009; Liu, 2010).
But is electrospinning applicable to fabricating the prostheses for blood vessels of various diameters? The material used for vascular implants should meet definite requirements. This material has to retain its mechanical performance and size under various loads and be porous to allow nutrients to penetrate inside the matrix but to display a selectively permeable to prevent the penetration of blood cells, which circulate in the vessels.
Optimization of the compositions for the outer and inner layers of the vascular prosthesis is of great importance; this is necessary to maintain the cell population with a required phenotype. The inner surface of the vessel wall should be appropriate for endothelial cell adhesion (sticking) and proliferation, while the outer surface should provide for the development of muscle layer. Finally, the fabricated vessels should be convenient for handling and should not induce and mechanical irritation or inflammatory response.
The electrospun vessels meet all these requirements. In particular, they have a much smoother surface suitable for the formation of a cell layer, unlike the currently used braided, knitted, or porous polytetrafluoroethylene vascular prostheses.
Testing for stability
The electrospinning technology for the fabrication of vascular prostheses is relatively simple. A metal rod with a necessary diameter is used as a collector electrode onto which the fiber is settled. After the process is completed, a tube ready to be used as a vascular prosthesis is taken from the electrode. Such prostheses made of polycaprolactone, polydioxanone, silk, collagens, and elastin have already been tested in laboratory animals but have not reached the clinical practice level yet (Boland, 2004; Garg, 2011).
 In Russia, the Bakulev Center for Cardiovascular Surgery (Russian Academy of Medical Sciences, Moscow) and several Siberian institutions—Institute of Chemical Biology and Fundamental Medicine, Siberian Branch, Russian Academy of Sciences (Novosibirsk); Meshalkin Institute of Circulation Pathology (Novosibirsk) and Institute of Integrated Problems in Cardiovascular Diseases (Kemerovo), Siberian Branch, Russian Academy of Medical Sciences—are involved in the development and study of electrospun vascular prostheses.
In Russia, the Bakulev Center for Cardiovascular Surgery (Russian Academy of Medical Sciences, Moscow) and several Siberian institutions—Institute of Chemical Biology and Fundamental Medicine, Siberian Branch, Russian Academy of Sciences (Novosibirsk); Meshalkin Institute of Circulation Pathology (Novosibirsk) and Institute of Integrated Problems in Cardiovascular Diseases (Kemerovo), Siberian Branch, Russian Academy of Medical Sciences—are involved in the development and study of electrospun vascular prostheses.
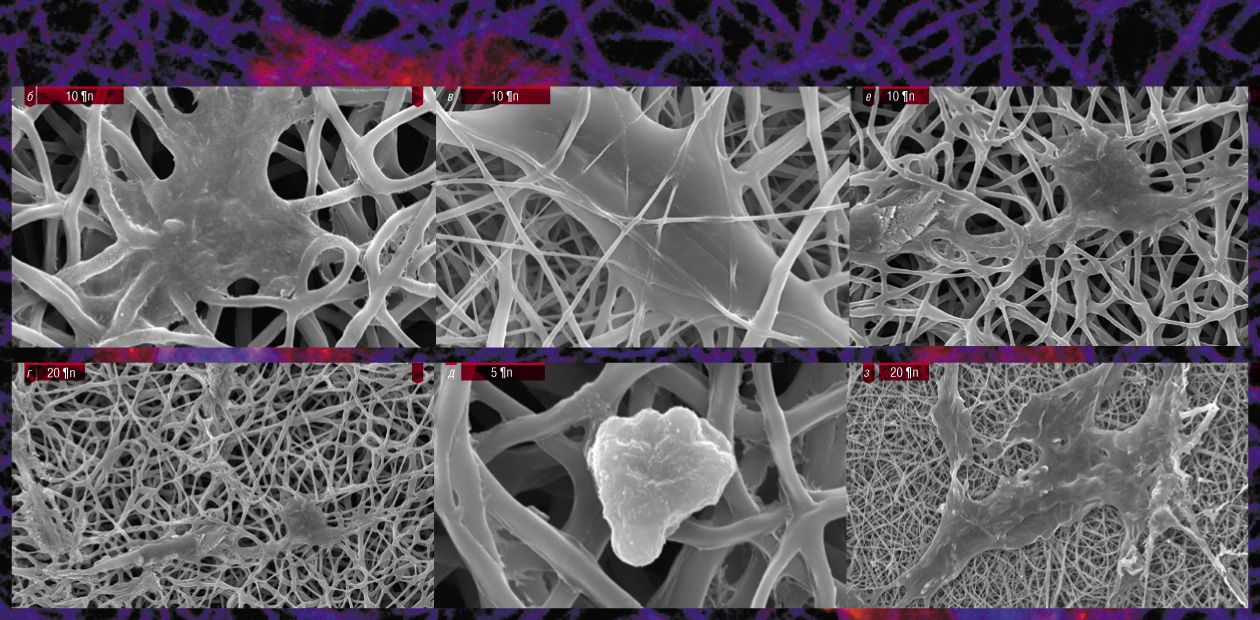
At the Institute of Chemical Biology and Fundamental Medicine, eight types of matrices from synthetic polymers (nylon, polycaprolactone, and polylactide-co-glycolide) or their mixtures with biological polymers have been tested for their ability to maintain cell adhesion and proliferation. Fibroblasts, a major cell type in the connective tissue, have appeared to stick well to all tested polymeric matrices and even to able to multiply on the nylon matrix without any additional growth factors. Endothelial cells, lining the inner surface of vessels, are more sensitive to the type of material: they reproduce well on the matrix of polylactide-co-glycolide supplemented with gelatin and growth factors.
There are strict requirements to the mechanical performance of vascular prostheses. These prostheses should endure a long-term cyclic stress, analogous to that in the body, be resistant to constriction and kinking, and fit well the recipient vascular walls.
Evidently, the tolerance to such stress combined with the retention of shape and size can be attained only when the corresponding stress falls within the range of elastic deformations. This means that after the load is removed, the material reverts to its original state without any residual deformation.
The mechanical properties of vascular prostheses were examined at the joint access center for mechanics with the Lavrentiev Institute of Hydrodynamics, Siberian Branch, Russian Academy of Sciences (Novosibirsk). The testing device comprising a reservoir with excess pressure and two PC-controlled valves whose successive mode simulated the blood pressure was produced for the dynamical tests of vascular prostheses. The nylon prostheses demonstrate a complete absence of any residual deformation after 100,000 load cycles with a change in pressure of 150 mm Hg.
Another important characteristic of vascular prostheses to be sewed into the recipient’s vessel is their stability to tearing with a thread. The conducted tests have demonstrated that this characteristic also meets all the requirements and amounts to 260 kgf. The degree of porosity of the tested prostheses, determining the blood loss, also complied with the requirements; this parameter was estimated according to water leakage through the wall at an excess pressure corresponding to the normal human upper blood tension.
A high resistance to constriction and kinking in the vascular prostheses of a large diameter is usually attained by pleating. However, this technical solution is inappropriate for small-diameter prostheses, since folds may contain dead circulation zones, thereby leading to inflammation and stenosis.
In order to increase the mechanical performance and stiffness of vascular prostheses, Novosibirsk researchers exposed them to high-energy electron beam radiation in an ILU-6 accelerator at the Budker Institute of Nuclear Physics, Siberian Branch, Russian Academy of Sciences. As is known, irradiation of materials induces chemical modifications, thereby altering their physicomechanical properties. In this case, exposure of polycaprolactone and nylon items to a dose of 25–200 kGy (for comparison, a typical dose used for sterilization of foodstuff does not exceed 30 kGy) increases the elastic modulus of these materials and, correspondingly, their stiffness. If a prosthesis is irradiated using a mask with a through pattern of radiation absorbing material, it is possible to form separate sites with elevated stiffness within the prosthesis, thereby avoiding vessel constriction.
Thus, all the tests of the polymeric matrices that Novosibirsk specialists produced by electrospinning have proved that they are potentially appropriate for manufacturing vascular prostheses. However, only the transplantation of a prosthesis into a living organism can give reliable information about its behavior in the blood channel.
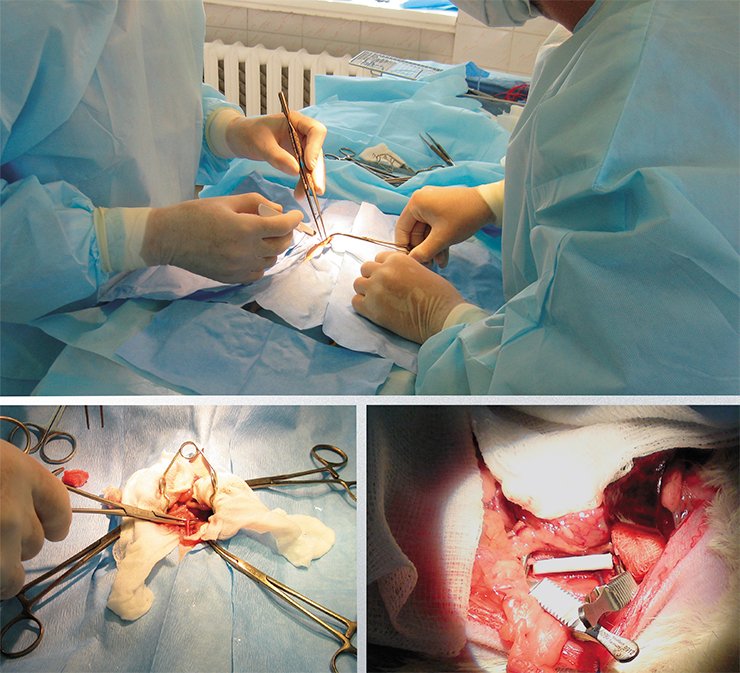
Prothesing of the ventral aorta in Wistar laboratory rats is a commonly used model for such a study. Prosthesis repair of a vessel with a diameter of 1.5–2.0 mm is not an easy task even for an expert. Surgeons from the Meshalkin Institute of Circulation Pathology completed the mission successfully: during the substitution of the ventral aorta with a polycaprolactone prosthesis, they put 13 (!) stitches to connect the ends of a tiny 2-mm prosthesis and even thinner vessel.
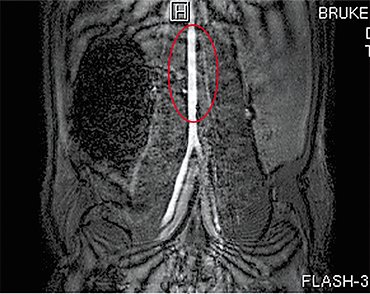 After the intervention, the animal quickly recovered, suggesting a normal performance of the vascular prosthesis. This was also confirmed by objective intravital examinations using magnetic resonance tomography at the SPF vivarium with the Institute of Cytology and Genetics, Siberian Branch, Russian Academy of Sciences (Novosibirsk). According to magnetic resonance angiography, neither stenosis nor aneurisms were present in the prosthesis and the blood circulation rate was the same as in a normal rat ventral aorta.
After the intervention, the animal quickly recovered, suggesting a normal performance of the vascular prosthesis. This was also confirmed by objective intravital examinations using magnetic resonance tomography at the SPF vivarium with the Institute of Cytology and Genetics, Siberian Branch, Russian Academy of Sciences (Novosibirsk). According to magnetic resonance angiography, neither stenosis nor aneurisms were present in the prosthesis and the blood circulation rate was the same as in a normal rat ventral aorta.
In addition to vascular implants, Novosibirsk researchers used electrospinning to produce a composite prosthesis of a hyaline cartilage; this prosthesis is a “stack” of sheets for the cultivation of chondroblasts. Such a stack can be fastened at an injured site with the help of a nontoxic photopolymerized gel.
As for the vascular prostheses, a new promising research field is evident here. Scientists plan to populate polymeric matrices with endothelial cells of the future recipient, which, in turn, will be produced from his/her fibroblasts via the stage of undifferentiated stem cells… But this is the topic for future papers.
References
Filatov I. Yu., Filatov Yu. N., Yakushkin M. S. Elektroformovanie voloknistykh materialov na osnove polimernykh mikro- i nanovolokon. Istoriya, teoriya, tekhnologiya, primenenie // Vestn. MITKhT im. M. V. Lomonosova. 2008. T. 3 (5). C. 3—18.